EFFICACY DATA
FOR ULCERATIVE
COLITIS
Proven to Give Patients a Chance at Early Symptom Relief and Endoscopic Control1

STUDY DESIGN
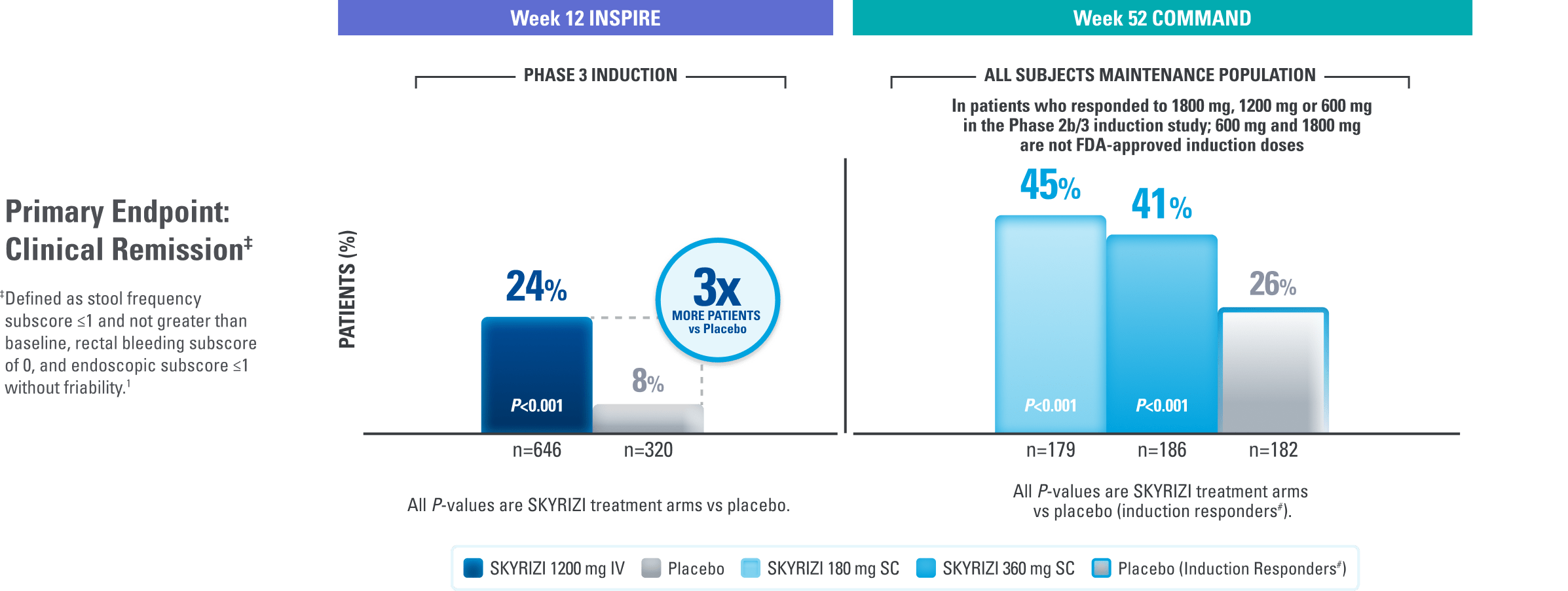
INSPIRE Induction (N=966) was a Phase 3, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of SKYRIZI 1200 mg IV, at Weeks 0, 4, and 8 vs placebo over 12 weeks in adult patients with moderate to severe UC. The primary endpoint was clinical remission‡ at Week 12.1
COMMAND Maintenance (N=547) was a Phase 3, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of SKYRIZI 180 mg or 360 mg SC vs placebo up to 52 weeks in adult patients who were clinical responders to one of three SKYRIZI induction regimens consisting of 600 mg, 1200 mg, or 1800 mg IV (the 600 mg and 1800 mg IV are not FDA-approved induction regimens) in the INSPIRE Phase 2b or Phase 3 studies. The primary endpoint was clinical remission‡ at Week 52. COMMAND also included a prespecified subgroup analysis inclusive only of patients who responded to the FDA-approved induction dosing of SKYRIZI 1200 mg IV (N=271).1
Patients Achieved Durable Remission at Weeks 12 and 521
Use the lowest effective dosage to maintain therapeutic response.
†Clinical response at Week 4 in UC per partial modified Mayo Score is a composite of Mayo stool frequency and rectal bleeding subscores and was defined as a decrease in total score ≥30% and ≥1 point from baseline and a decrease in rectal bleeding subscore ≥1 or rectal bleeding subscore of 0 or 1.1
§Endoscopic improvement in UC is defined as Mayo endoscopic subscore of 0 or 1 without friability.1 Endoscopic results are based on a full colonoscopy or flexible sigmoidoscopy, at the discretion of the investigator.2
||Endoscopic remission in UC is defined as Mayo endoscopic subscore of 0.1 Endoscopic results are based on a full colonoscopy or flexible sigmoidoscopy, at the discretion of the investigator.2
¶Steroid-free clinical remission in UC is defined as clinical remission per modified Mayo Score (rectal bleeding, stool frequency, endoscopic subscore) at Week 52 and corticosteroid-free for ≥90 days immediately preceding Week 52.
#Patients who achieved clinical response with SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.2
IV=intravenous; SC=subcutaneous.
EARLY SYMPTOM RELIEF
Relief of Rectal Bleeding and Stool Frequency as Early as Week 41,3
Clinical Response* at Weeks 4, 8, 12
(Composite of rectal bleeding and stool frequency subscores)
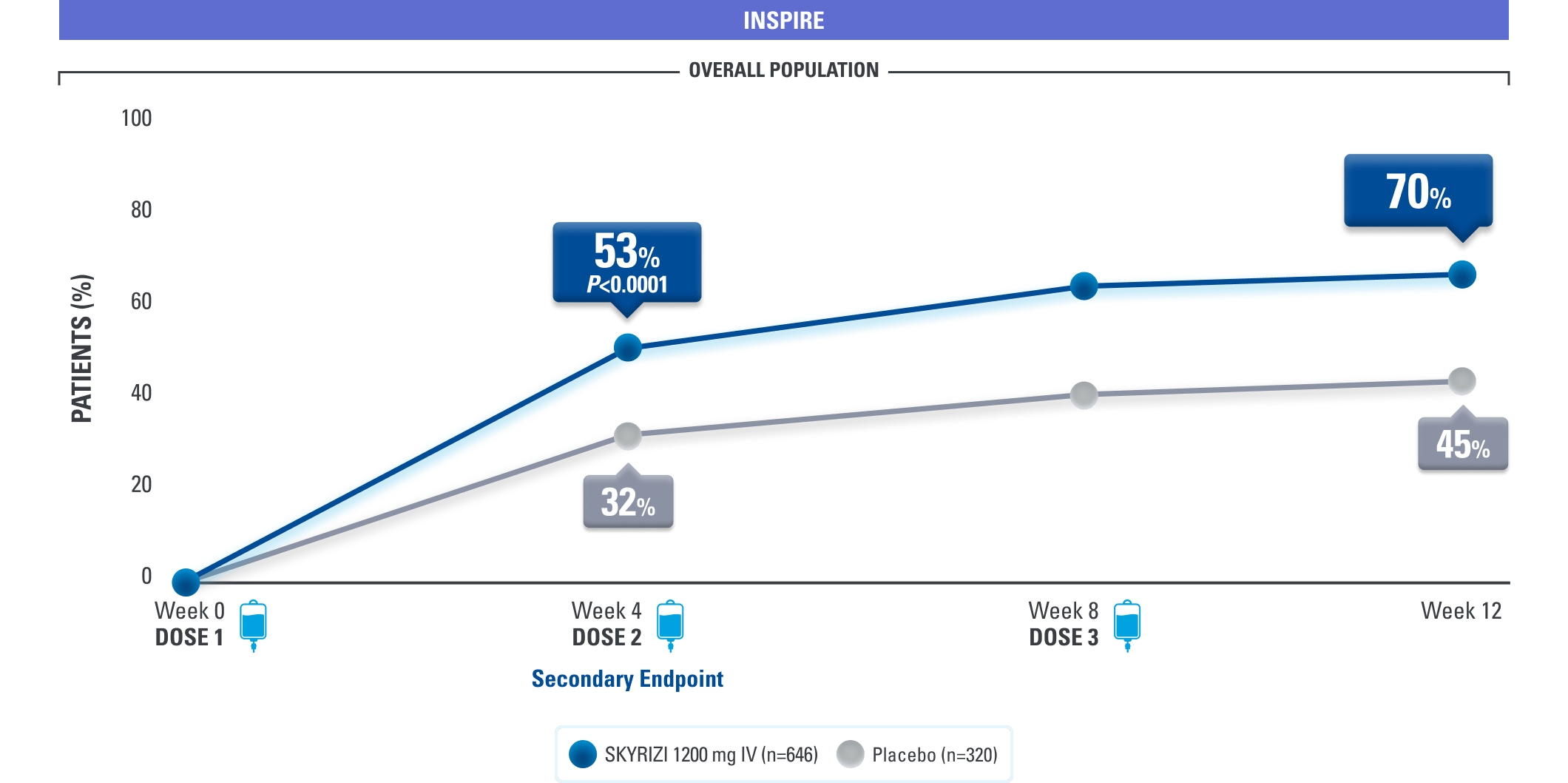
DATA LIMITATION: Clinical response at Weeks 8 and 12 were prespecified, nonranked endpoints not controlled for multiplicity. No conclusions or statistical inferences can be made.
All P-values are SKYRIZI treatment arms vs placebo.
*Clinical response at Week 4 in UC per partial modified Mayo Score is a composite of Mayo stool frequency and rectal bleeding subscores and was defined as a decrease in total score ≥30% and ≥1 point from baseline and a decrease in rectal bleeding subscore ≥1 or rectal bleeding subscore of 0 or 1.1
IV=intravenous.
ENDOSCOPIC CONTROL
Endoscopic Control: Visible Improvement of the Colon Lining at 1 Year1,3
Endoscopic Improvement* at Week 521,3
(Endoscopy subscore of 0 or 1, without friability)
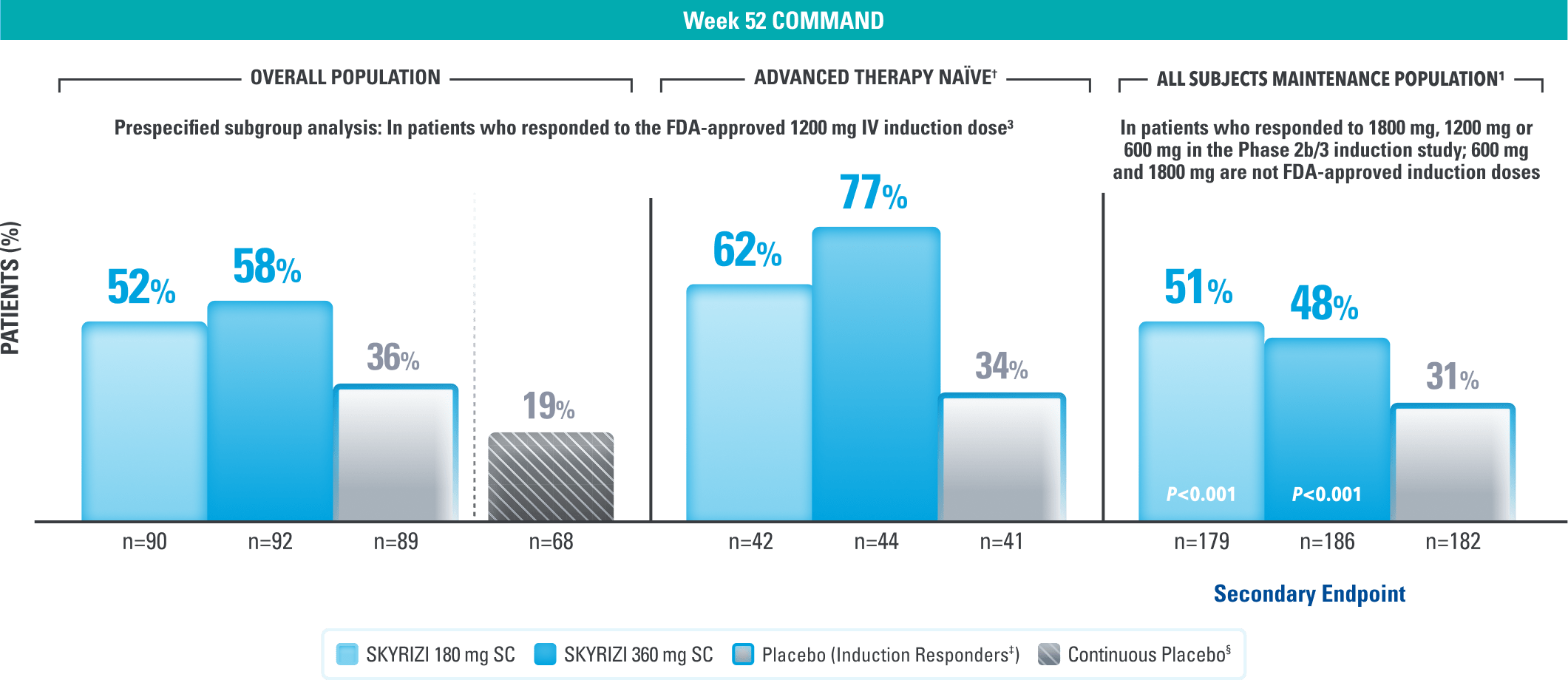
Continuous placebo data not intended for direct comparison.
LIMITATIONS OF PRESPECIFIED SUBGROUP ANALYSIS: Results shown are only among patients who responded to the FDA-approved 1200 mg IV induction dose. Excluded are those who received unapproved induction doses (600 mg or 1800 mg). Data were not controlled for multiplicity. No conclusions or statistical inferences can be made.3
All P-values are SKYRIZI treatment arms vs placebo (induction responders‡).
†Advanced therapy-naïve patients include some patients who were exposed to an advanced therapy (biologics, JAK inhibitors, and/or S1P receptor modulators) but did not experience treatment failure.1
*Endoscopic improvement in UC is defined as Mayo endoscopic subscore of 0 or 1 without friability.1 Endoscopic results are based on a full colonoscopy or flexible sigmoidoscopy, at the discretion of the investigator.2
‡Patients who achieved clinical response with SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.2
§Patients who responded to placebo in induction were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.
IV=intravenous; JAK=Janus kinase; SC=subcutaneous; S1P=Sphingosine-1-phosphate.
Endoscopic Control at 1 Year3
Endoscopic Remission* at Week 523
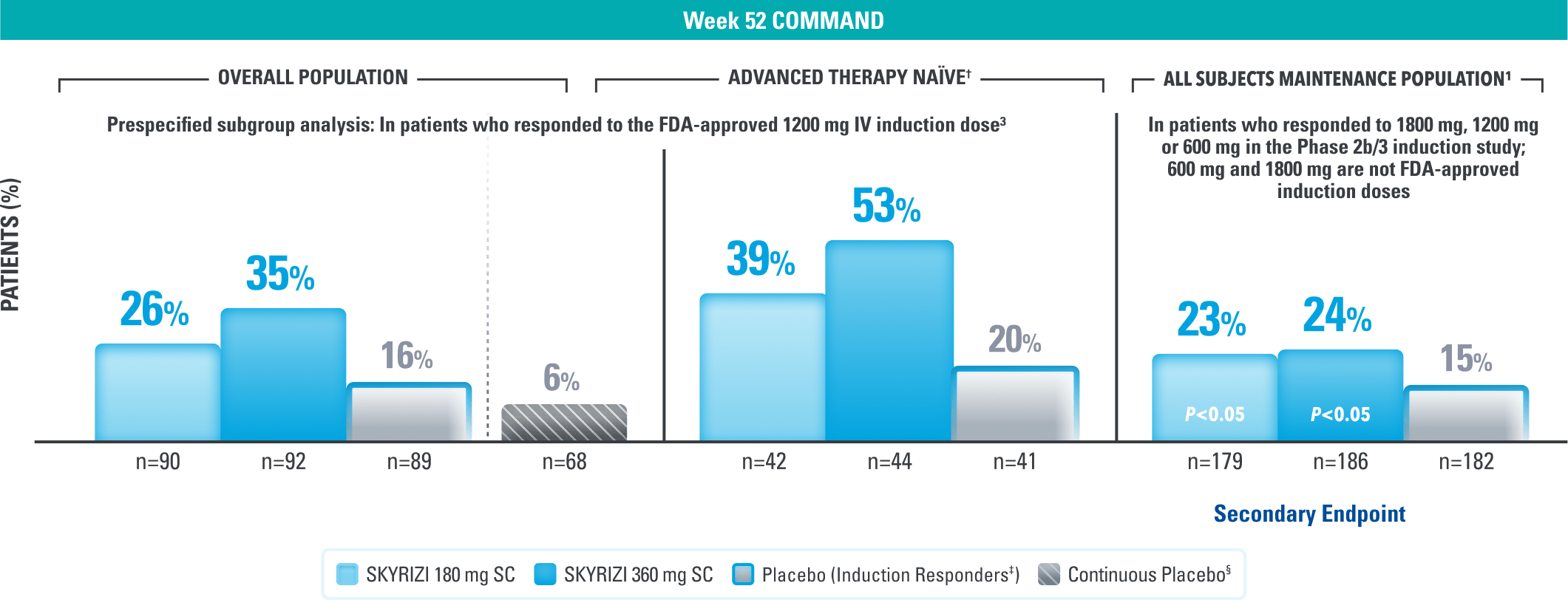
Continuous placebo data not intended for direct comparison.
LIMITATIONS OF PRESPECIFIED SUBGROUP ANALYSIS: Results shown are only among patients who responded to the FDA-approved 1200 mg IV induction dose. Excluded are those who received unapproved induction doses (600 mg or 1800 mg). Data were not controlled for multiplicity. No conclusions or statistical inferences can be made.3
All P-values are SKYRIZI treatment arms vs placebo (induction responders‡).
†Advanced therapy-naïve patients include some patients who were exposed to an advanced therapy (biologics, JAK inhibitors, and/or S1P receptor modulators) but did not experience treatment failure.1
*Endoscopic remission in UC is defined as Mayo endoscopic subscore of 0.1 Endoscopic results are based on a full colonoscopy or flexible sigmoidoscopy, at the discretion of the investigator.2
‡Patients who achieved clinical response with SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.2
§Patients who responded to placebo in induction were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.
IV=intravenous; JAK=Janus kinase; SC=subcutaneous; S1P=Sphingosine-1-phosphate.
DURABLE REMISSION
Durable Remission at 1 Year3
Clinical Remission* at Week 523
(Composite of rectal bleeding, stool frequency, and endoscopy subscores)
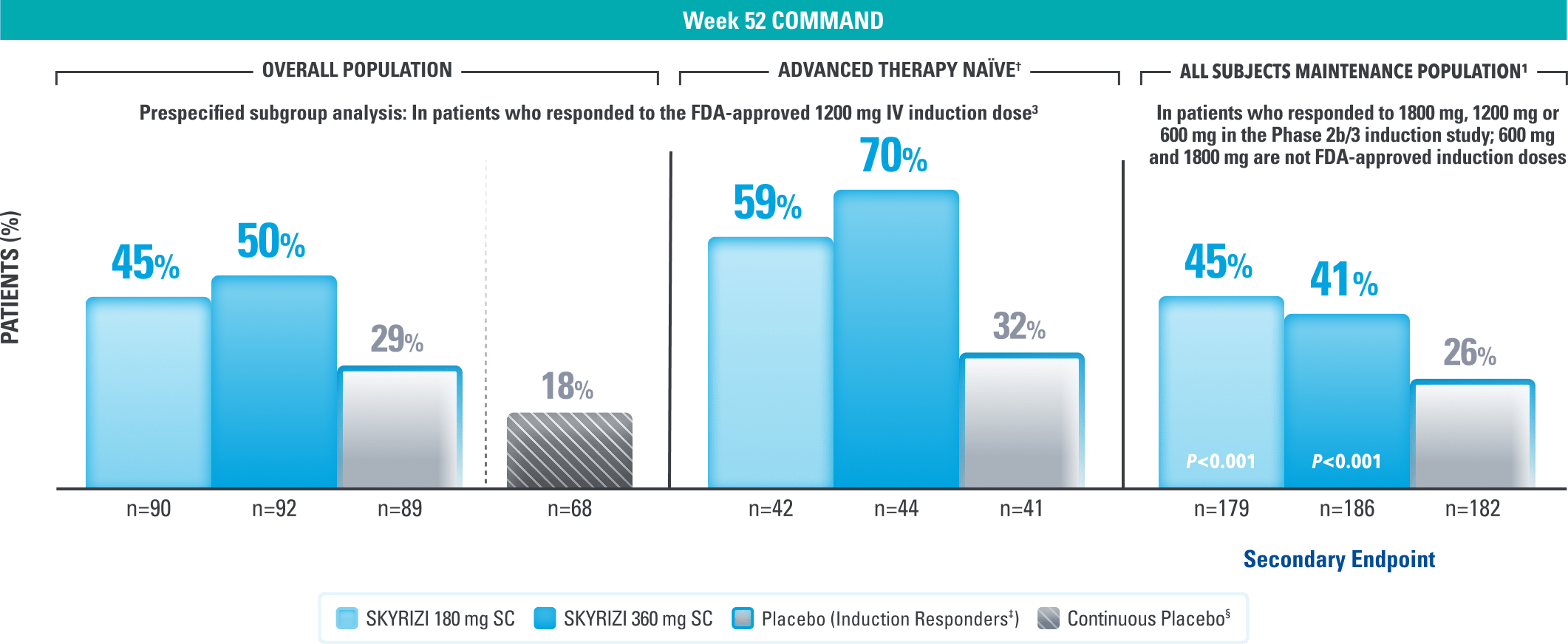
Continuous placebo data not intended for direct comparison.
LIMITATIONS OF PRESPECIFIED SUBGROUP ANALYSIS: Results shown are only among patients who responded to the FDA-approved 1200 mg IV induction dose. Excluded are those who received unapproved induction doses (600 mg or 1800 mg). Data were not controlled for multiplicity. No conclusions or statistical inferences can be made.
All P-values are SKYRIZI treatment arms vs placebo.
In a post hoc analysis of patients who responded to the FDA-approved 1200 mg IV induction dose, among those in clinical remission at Week 52,
>97% were also corticosteroid-free for ≥90 days prior to visit.5
(180 mg [N=39/40]; 360 mg [N=44/45]; placebo [induction responders‡] [N=26/26])
POST HOC LIMITATION: Data were not controlled for multiplicity. No conclusions or statistical inferences can be made.
At Week 52, in the prespecified subgroup analysis in patients who responded to the FDA-approved 1200 mg IV induction dose, 44% of patients on SKYRIZI 180 mg and 48% of patients on SKYRIZI 360 mg achieved corticosteroid-free clinical remission|| compared to 29% on placebo.3
†Advanced therapy-naïve patients include some patients who were exposed to an advanced therapy (biologics, JAK inhibitors, and/or S1P receptor modulators) but did not experience treatment failure.1
*Clinical remission in UC is defined as stool frequency subscore ≤1 and not greater than baseline, rectal bleeding subscore of 0, and endoscopic subscore ≤1 without friability.1
‡Patients who achieved clinical response with SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.2
§Patients who responded to placebo in induction were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.4
IISteroid-free clinical remission in UC is defined as clinical remission per modified Mayo Score (rectal bleeding, stool frequency, endoscopic subscore) at Week 52 and corticosteroid-free for ≥90 days immediately preceding Week 52.1
IV=intravenous; JAK=Janus kinase; SC=subcutaneous; S1P=Sphingosine-1-phosphate.
Steroid-free Remission Data at 1 Year1,3
Corticosteroid-free Clinical Remission* at Week 521,3
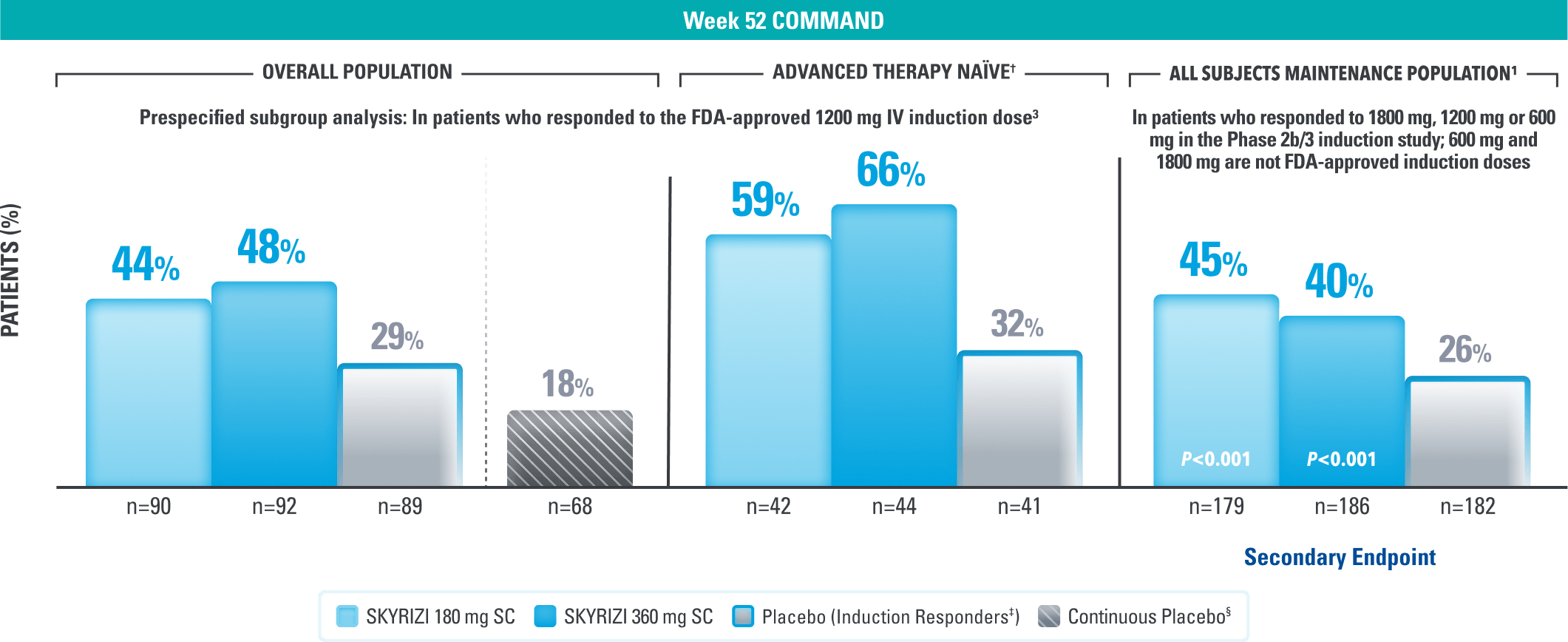
Continuous placebo data not intended for direct comparison.
LIMITATIONS OF PRESPECIFIED SUBGROUP ANALYSIS: Results shown are only among patients who responded to the FDA-approved 1200 mg IV induction dose. Excluded are those who received unapproved induction doses (600 mg or 1800 mg). Data were not controlled for multiplicity. No conclusions or statistical inferences can be made.
All P-values are SKYRIZI treatment arms vs placebo.
†Advanced therapy-naïve patients include some patients who were exposed to an advanced therapy (biologics, JAK inhibitors, and/or S1P receptor modulators) but did not experience treatment failure.1
*Steroid-free clinical remission in UC is defined as clinical remission per modified Mayo Score (rectal bleeding, stool frequency, endoscopic subscore) at Week 52 and corticosteroid-free for ≥90 days immediately preceding Week 52.1
‡Patients who achieved clinical response with SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.2
§Patients who responded to placebo in induction were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.4
IV=intravenous; JAK=Janus kinase; SC=subcutaneous; S1P=Sphingosine-1-phosphate.
Recommended for You
SKYRIZI Safety
SKYRIZI Dosing
Skyrizi Complete



