“When I’m using the OBI, I sometimes answer email, scroll through social media, or even walk around the house or get the mail.”
– Jonathan, a real SKYRIZI patient
ON-BODY
INJECTOR
(OBI)
A Device Designed With Your Patients in Mind
Single-dose SC injector with SKYRIZI prefilled cartridge
Get to Know the SKYRIZI OBI


Compact Size
Small enough to fit in the palm of the hand


Hands-Free
While adhered to the abdomen or thigh and after activation, patients can do moderate physical activities such as walking, reaching, and bending


Hidden Needle
Discreet injection so patients don’t see the needle
Please see the Instructions for Use to provide patients and/or caregivers proper training on the OBI.
SC=subcutaneous.
REAL-WORLD OBI DATA
Backed by Real-World Experience: What do real patients think of the OBI?
The ASPIRE-Crohn’s Disease Study: Assessing the real-world experience of patients with moderately to severely active Crohn’s taking SKYRIZI2
In a questionnaire, Crohn’s patients reported their experiences with the OBI at Week 12

LIMITATIONS: The survey data were collected from patients who enrolled in the Skyrizi Complete patient support program, and these patients may have different perceptions of the SKYRIZI OBI vs those who did not enroll. The questions in this interim analysis were not based on a standardized validated tool for Crohn’s disease. As-observed data may bias results. There was also potential for recall bias because outcomes were self-reported. Safety and tolerability data were not collected.
ASPIRE-CROHN’S DISEASE STUDY DESIGN: Based on a prospective, longitudinal patient survey of 312 adult patients with moderate to severe Crohn’s disease who had not been treated with SKYRIZI previously and chose to opt in to the study. Surveys were completed at baseline (prior to first SKYRIZI infusion) and at Weeks 2, 4, 12, 20, 28, 36, 44, and 52 after initiation of SKYRIZI. Data are from an interim analysis of patients who had completed surveys at 12 weeks (n=187). Patients reported their experience with the OBI through questions on ease of learning, ease of use, and satisfaction using a 5-point Likert scale that ranged from “very difficult/dissatisfied” to “very easy/satisfied.”
ADMINISTRATION CONSIDERATIONS: SKYRIZI is intended for use under the guidance and supervision of a healthcare professional (HCP). SKYRIZI vial for intravenous administration is intended for administration by an HCP. Prior to starting therapy, please refer to the Dosage and Administration section of the Prescribing Information for complete information on how to initiate, prepare, and administer SKYRIZI. Patients may self-inject SKYRIZI using the on-body injector with prefilled cartridge after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI according to the Instructions for Use.1
PATIENT SATISFACTION DATA
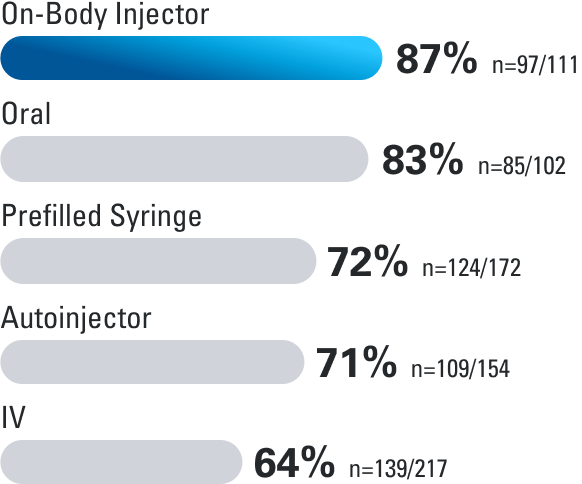
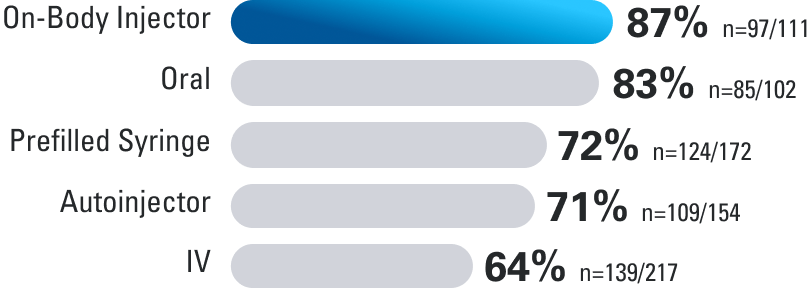
Patient Satisfaction Rates
In a patient-reported survey, a questionnaire was completed by 352 adult patients with moderate to severe Crohn’s disease or ulcerative colitis, who reported their experiences with 1 or more routes of administration for maintenance treatment.3
Route of administration during maintenance satisfaction rates
How satisfied are you with the following route(s) of admnistraton for delivering the maintenance therapy?
Percentage of patients who reported “satisfied” or “highly satisfied”
OBI 8-week maintenance dosing schedule satisfaction rates
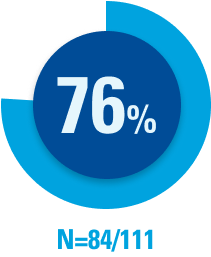
Of those patients with experience using the SKYRIZI OBI, 76% (N=84/111) reported they “strongly agree/agree” that the 8-week maintenance dosing schedule fits better with their routine than a treatment option with a monthly injection schedule.
LIMITATIONS: There are no head-to-head studies comparing routes of administration in IBD so no statistical or clinical conclusions can be drawn. These questions in this interim analysis were not based on a standardized validated tool for IBD. There was also potential for recall bias because outcomes were self-reported. The data from this questionnaire may not be reflective of all patients who have used these routes of administration for IBD treatment. Efficacy and safety data were not collected.
This survey only included patients who received SKYRIZI maintenance dosing via OBI. This graphic is not intended to compare the efficacy or safety of IBD treatments using these routes of administration. Additional considerations, aside from route of administration, should be taken into account by the HCP and patient before making a treatment decision. Refer to the full prescribing information for more details on approved IBD maintenance treatments.
PATIENT QUESTIONNAIRE STUDY DESIGN: Prospective, ongoing patient survey conducted in the US between December 2024 and February 2025 wherein 307 adult patients self-reported outcomes including diagnosis of moderate-to-severe CD or UC at least 1 year prior to taking the survey and use of FDA-approved prescription medication along with the approved route of administration for CD or UC. 102 adult patients on SKYRIZI reported receiving induction (IV) and maintenance (SC) dosing per prescribing information and appropriate training on the OBI from an HCP. Patients were required to either be currently taking or have previously taken maintenance dosing for IBD treatment via at least one of the routes of administration (4 months of treatment with SKYRIZI 180 mg SC or 360 mg SC Q8W or 6 months of treatment with all other medications). Patients reported their experience with the routes of administration on IBD treatment using a 5-point Likert scale that ranged from “not satisfied at all/less satisfied” to “highly satisfied/satisfied”.
IBD=inflammatory bowel disease; IL-23i=interleukin-23 inhibitor; IV=intravenous; OBI=on-body injector; Q4W=every 4 weeks; Q8W=every 8 weeks; SC=subcutaneous.



Recommended for You
SKYRIZI Access
Support for Your Patients and Practice



