SEQUENCE CROHN'S H2H DATA
Results from a head-to-head study
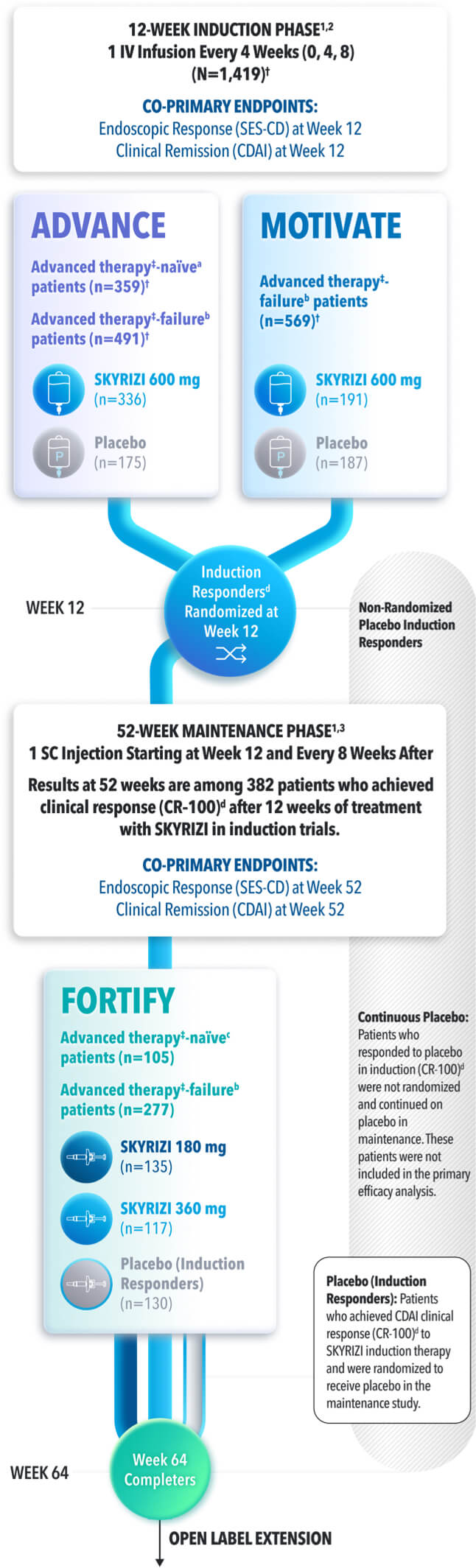
Endpoints in ADVANCE, MOTIVATE and FORTIFY1
All p-values are SKYRIZI treatment arms vs. placebo.
Placebo (Induction Responders): Patients who achieved CDAI clinical response (CR-100)‡ to SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.
STUDY DESIGNS
ADVANCE (N=850) and MOTIVATE (N=569) Induction studies were 12-week, randomized, double-blind, placebo-controlled studies that evaluated the efficacy and safety of SKYRIZI in patients with moderately to severely active Crohn’s disease§ who demonstrated prior treatment failure to conventional and/or biologic treatment.4 Patients received an IV infusion of SKYRIZI 600 mg (recommended dose), risankizumab-rzaa 1200 mg|| or placebo at Weeks 0, 4, and 8.1,2
FORTIFY (N=382) Maintenance study was a 52-week study that evaluated the efficacy and safety of SKYRIZI in patients who achieved clinical response (decrease in CDAI ≥100)‡ from SKYRIZI induction in the ADVANCE and MOTIVATE studies. Patients were randomized to SKYRIZI 180 mg SC, SKYRIZI 360 mg SC, or placebo at Week 12 and every 8 weeks thereafter.1,3
Endoscopic Response: Defined as decrease in SES-CD >50% from baseline, or a decrease of at least 2 points for subjects with a baseline score of 4 and isolated ileal disease, based on central reading. The sections evaluated on endoscopy are the: rectum, sigmoid and left colon, transverse colon, right colon and ileum (per SES-CD assessment).
Clinical Remission: Defined as a CDAI score <150 points.
Endoscopic Remission: Defined as SES-CD ≤4 and at least a 2-point reduction versus baseline and no subscore greater than 1 in any individual variable, as scored by a central reviewer.
*The mixed population includes patients who had inadequate response, loss of response, or intolerance to one or more biologics (biologic failure), as well as patients who had never demonstrated inadequate response, loss of response, or intolerance to a biologic (bio-naïve; includes 13% in ADVANCE and 8% in FORTIFY who were bio-exposed).
†Prior biologic failure includes inadequate response, loss of response, or intolerance to one or more biologics.1
‡Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
§Defined by CDAI of 220 to 450, an average daily SF ≥4 and/or average daily APS ≥2, and a SES-CD of ≥6, or ≥4 for isolated ileal disease, excluding the narrowing component.
||The 1200 mg IV induction dose did not demonstrate additional treatment benefit over the 600 mg IV dose and is not a recommended regimen.
APS=abdominal pain score; AT=advanced therapy; CDAI=Crohn's disease activity index; IV=intravenous; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease; SF=stool frequency.
Crohn’s Disease Therapies Evaluated in an Open-Label Assessor Blinded Head-to-Head Trial4,5

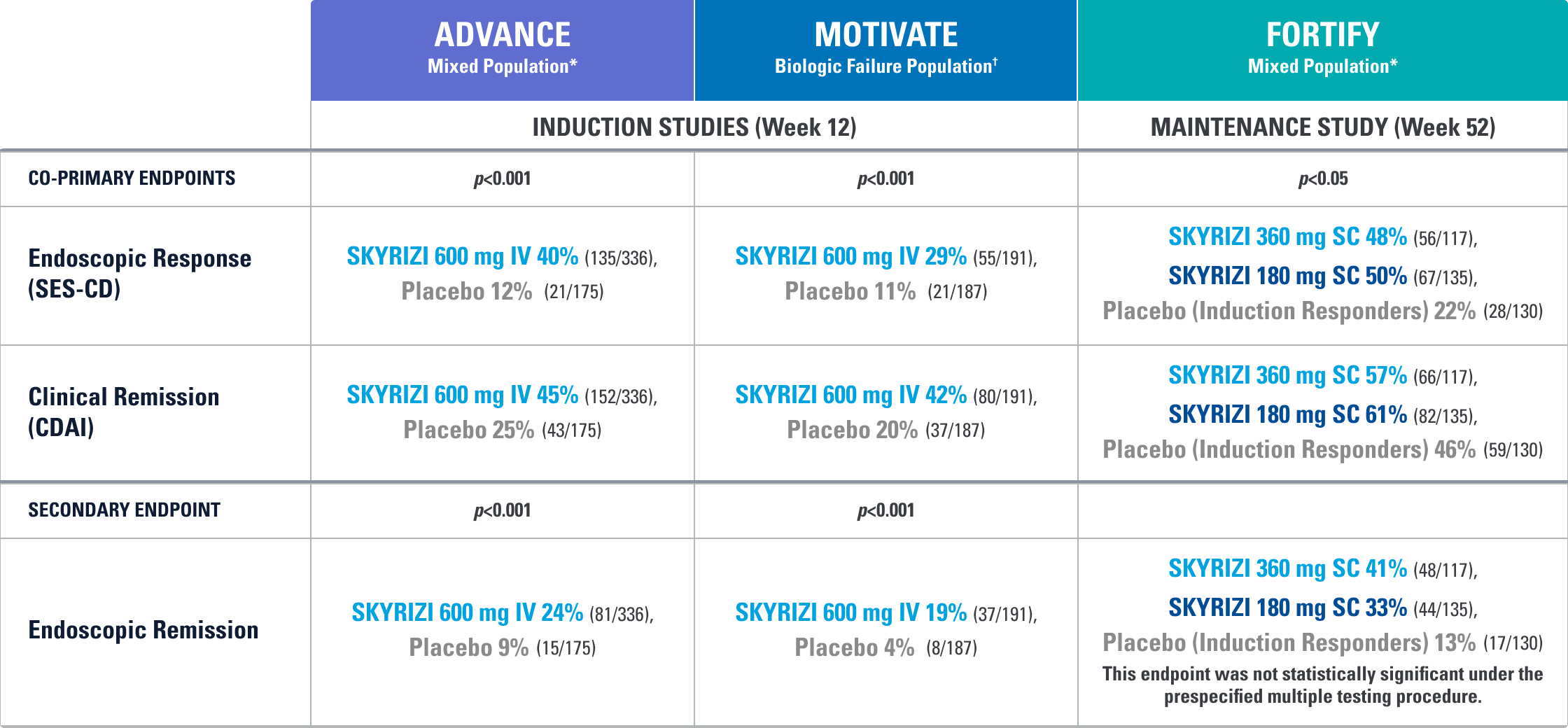
PRIMARY ENDPOINTS EVALUATED:
- Non-Inferiority Endpoint: CLINICAL REMISSION (CDAI <150) AT WEEK 24*
- Superiority Endpoint: ENDOSCOPIC REMISSION† (SES-CD ≤4) AT WEEK 48‡
*The percentage of subjects who met clinical remission at Week 24. Statistical requirement for non-inferiority achieved with ~50% of study population.
†Endoscopic remission: SES-CD ≤4 and at least a 2-point reduction versus baseline and no subscore greater than 1 in any individual variable, as scored by a central reviewer.
‡The percentage of subjects who met endoscopic remission at Week 48.
Study Design Intro: SEQUENCE was a Phase 3, multicenter, randomized, open-label, efficacy assessment-blinded§ study of SKYRIZI (n=255) compared to STELARA® (ustekinumab)|| (n=265) for the treatment of adult patients with moderate to severe Crohn’s disease who have failed anti-TNF therapy. Eligible patients were randomized (1:1) to receive either SKYRIZI (600 mg IV to 360 mg SC) or STELARA® (weight-based¶ IV to 90 mg SC). After induction dosing was completed, patients remained on their respective therapy throughout the duration of the maintenance period (treat-through study design). No dose escalation allowed throughout the trial.
DOSING: The lowest effective dosage for SKYRIZI should be used to maintain therapeutic response. The comparative effectiveness of SKYRIZI 180 mg is unknown, as it was not evaluated in this study.
§The investigator and site personnel were blinded to the results of the clinical outcomes (CDAI) and endoscopies were centrally read with assessors blinded to study drug.
||Active Comparator: 31 patients received US-approved ustekinumab. All other patients received European Union-approved ustekinumab. The comparability between US and non-US approved ustekinumab has not been established.
¶Baseline Stelara IV dose is weight based: ≤55 kg: 260 mg; >55 kg to 85 kg: 390 mg; or >85 kg: 520 mg.
STELARA® is a registered trademark of Johnson & Johnson. See US Prescribing Information for further information.
Non-inferiority Endpoint: This primary endpoint was measured in ~50% of total population. This measure was based on a non-inferiority margin of 10% at the 0.05, 2-sided significance level, where a margin of 10% was selected based on Physicians’ perspective on the clinical meaningfulness of inflammatory bowel disease trial results: an International Organization for the Study of Inflammatory Bowel Disease (IOIBD) survey.4
Superiority Endpoint: This primary endpoint was evaluated based on a 0.05, 2-sided significance level.
NRI-MI: Non-responder imputation for missing data with the exception that if the reason for missing data is due to COVID-19 infection or logistical restriction due to pandemic or geopolitical conflict, the patient's assessment will be imputed using multiple imputation.
CDAI=Crohn's disease activity index; IV=intravenous; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease.
H2H ENDPOINTS
All Primary and Ranked Secondary Endpoints Met4,5
SEQUENCE was a Phase 3, open-label, head-to-head study comparing SKYRIZI to STELARA® (ustekinumab) conducted entirely in adult patients with moderate to severe Crohn’s disease who have failed anti-TNF therapy
DATA LIMITATION: The open-label nature of this study may have introduced bias and influenced these results.
Endoscopic remission outcomes were unavailable for 12.9% (33/255) for SKYRIZI patients and 26% (69/265) for STELARA® patients.6
DOSING: The lowest effective dosage for SKYRIZI should be used to maintain therapeutic response. The comparative effectiveness of SKYRIZI 180 mg is unknown, as it was not evaluated in this study.
The investigator and site personnel were blinded to the results of the clinical outcomes (CDAI) and endoscopies were centrally read with assessors blinded to the study drug.4
Non-inferiority Endpoint: This primary endpoint was measured in ~50% of total population. This measure was based on a non-inferiority margin of 10% at the 0.05, 2-sided significance level, where a margin of 10% was selected based on Physicians’ perspective on the clinical meaningfulness of inflammatory bowel disease trial results: an International Organization for the Study of Inflammatory Bowel Disease (IOIBD) survey.4
Steroid-free Endoscopic Remission: Total population of patients who achieved endoscopic remission, defined as SES-CD ≤4 and at least a 2-point reduction versus baseline and no subscore greater than 1 in any individual variable, as scored by a central reviewer, who also did not receive a steroid at Week 48.5
Clinical Remission: CDAI <150.5
Endoscopic Remission: SES-CD ≤4 and at least a 2-point reduction vs baseline and no subscore >1 in any individual variable, as scored by a central reviewer.5
Endoscopic Response: Decrease in SED-CD >50% from baseline (or for participants with isolated ileal disease and a baseline SES-CD of 4, at least a 2-point reduction from baseline); the sections evaluated on endoscopy are the rectum, sigmoid and left colon, transverse colon, right colon, and ileum (per SES-CD assessment).5
Superiority Endpoint: Evaluated based on a 0.05, 2-sided significance level.4,5
*Active Comparator: 31 patients received US-approved ustekinumab. All other patients received European Union-approved ustekinumab. The comparability between US and non-US approved ustekinumab has not been established.
†Baseline STELARA® IV dose is weight based: ≤55 kg: 260 mg; >55 kg to 85 kg: 390 mg; or >85 kg: 520 mg.4
STELARA® is a registered trademark of Johnson & Johnson. See US Prescribing Information for further information.
CDAI=Crohn's disease activity index; H2H=head-to-head; IL-23i=interleukin-23 inhibitor; NRI-Ml=nonresponder imputation (NRI) while incorporating multiple imputation (MI) to handle missing data due to COVID-19 infection or logistical restrictions due to pandemic and/or geopolitical conflict; Q8W=every 8 weeks; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease; TNF=tumor necrosis factor.
POST HOC STEROIDS DATA
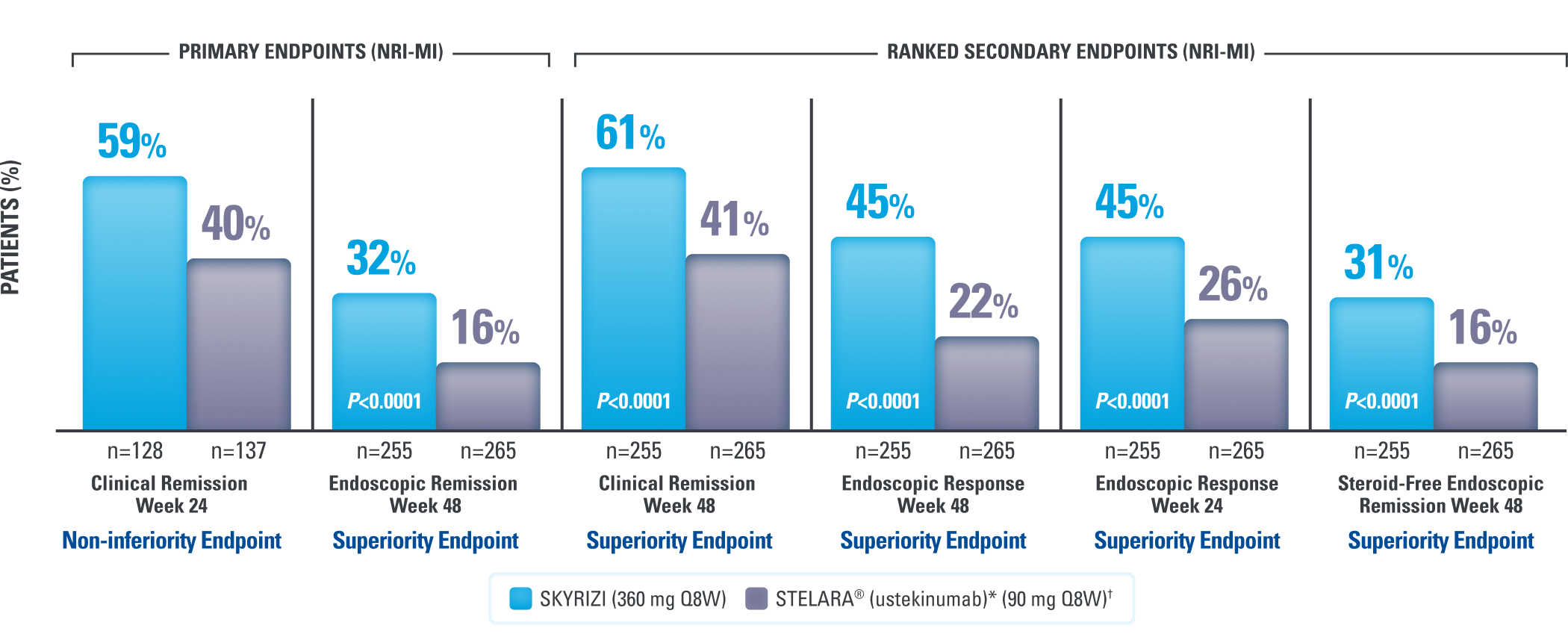
Post Hoc Analyses of SEQUENCE at Weeks 8, 24, 48, and 100
Steroid-Free* Data for SKYRIZI Patients8
Percent of Patients Who Discontinued Corticosteroids
Mean corticosteroid use was a prednisone-equivalent dose of 0.8 mg/day for SKYRIZI (n=51) and 3.6 mg/day for STELARA® (n=47) at Week 48.8
*Steroid-free is defined as patients who were not taking a corticosteroid at time point presented among patients taking a corticosteroid at baseline.8
All data were evaluated among patients on corticosteroids at baseline.9
At Week 100 in the Open-Label Extension (OLE) (as observed [AO]),
98% (44/45) of SKYRIZI patients who were taking corticosteroids at baseline were steroid-free9*
LIMITATIONS: The open-label nature of this study may have introduced bias and influenced these results. All post hoc analysis data including OLE were not controlled for multiplicity and no statistical or clinical inferences can be made. AO: A patient may not have had a response at each time point and any missing visit data were excluded from calculations for that visit, which may increase percent responders. All observed data were used regardless of premature discontinuation of study drug or initiation of concomitant medication.
DOSING: The lowest effective dosage for SKYRIZI should be used to maintain therapeutic response. The comparative effectiveness of SKYRIZI 180 mg is unknown, as it was not evaluated in this study.
SAFETY DATA
SEQUENCE Safety Profile: Evaluation Through Week 484,5
Treatment-Emergent Adverse Events Through Week 48
Swipe to scroll
| SKYRIZI (N=262, PYs=257.6) | STELARA® (ustekinumab)*(N=265, PYs=269.9) | |
|---|---|---|
| % (E/100 PYs) | % (E/100 PYs) | |
| Any AE | 85.1 (341.2) | 82.6 (282.7) |
| Serious AE | 10.3 (14.0) | 17.4 (23.7) |
| AE Leading to Discontinuation of Study Drug | 3.8 (3.9) | 4.9 (5.2) |
| Death | 0 | 0 |
| Infections | 53.8 (90.4) | 44.5 (62.6) |
| Serious Infection | 3.1 (3.9) | 4.2 (5.2) |
| Opportunistic Infection (Excluding TB/Herpes Zoster) | 0.4 (0.4) | 0 |
| Malignant Tumors | 0.4 (0.4) | 0.4 (0.4) |
| Adjudicated MACE | 0 | 0.4 (0.4) |
| Active TB | 0 | 0 |
| Herpes Zoster | 0.4 (0.4) | 0.4 (0.4) |
| Serious Hypersensitivity | 0 | 0 |
| Adjudicated Anaphylactic Reaction | 0 | 0 |
| Hepatic Events | 6.9 (10.1) | 5.3 (8.5) |
| Injection Site Reaction | 1.9 (1.9) | 2.3 (3.0) |

The safety profile of SKYRIZI was generally consistent with previously reported studies and as described in the full Prescribing Information5
The safety rates observed in clinical trials may not reflect clinical practice1
For the safety population, 7 patients randomized to SKYRIZI received 1200 mg IV and/or 180 mg SC and were included only in the safety analysis. Safety data presented includes all patients who received at least 1 dose of study drug. See US Prescribing Information for further information.
*The investigator and site personnel were blinded to the results of the clinical outcomes (CDAI) and endoscopies were centrally read with assessors blinded to study drug.
AE=adverse event; IV=intravenous; MACE=major adverse cardiovascular event; PYs=patient-years; SC=subcutaneous; TB=tuberculosis; TNF=tumor necrosis factor.
Recommended for You
Efficacy in Crohn’s Disease
SKYRIZI Dosing