SKYRIZI Is the #1 Prescribed Biologic
in Crohn’s Disease + Ulcerative Colitis 2*
*Source: Symphony Health, an ICON plc Company, IDV®; Total Combined Prescription Count from January 2025 to March 2025.2
US-MULT-250253
EXPERIENCE

SKYRIZI Is the #1 Prescribed Biologic
in Crohn’s Disease + Ulcerative Colitis 2*
*Source: Symphony Health, an ICON plc Company, IDV®; Total Combined Prescription Count from January 2025 to March 2025.2
EXPERIENCE

SKYRIZI Is the #1 Prescribed Biologic
in Crohn’s Disease + Ulcerative Colitis 2*
*Source: Symphony Health, an ICON plc Company, IDV®; Total Combined Prescription Count from January 2025 to March 2025.2

448,930
Patients Prescribed Worldwide
Since 2019 Starting With Ps4†
†Source: Symphony Health, an ICON plc Company, IDV®; September 1, 2016 to November 30, 2024.

69,794
Patients Prescribed in IBD in the US5†

‡"First-line" is defined as the first biologic prescription among patients who have not been prescribed biologics in the last 3 years. §Source: IQVIA (NPA, NSP) and Symphony Health data through February 2025.6

IISource: Integrated Symphony Health as of 3/2024.7
¶The term branded systemic is defined as systemic drugs that are sold by a specific name or trademark and protected by patent. OTC stands for over-the-counter medications.
IBD=inflammatory bowel disease; NPA=National Prescription Audit; NSP=National Sales Perspectives.
MECHANISM OF ACTION (MOA)
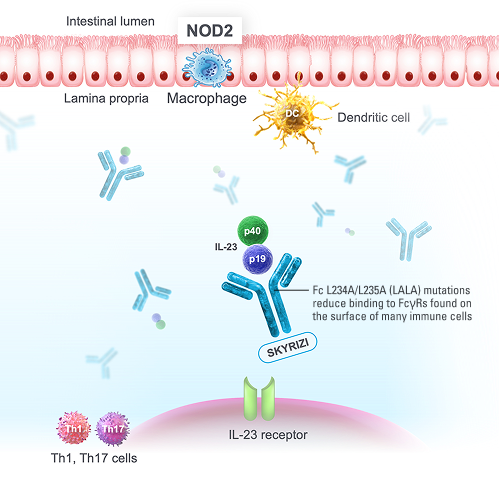
SKYRIZI is a humanized IgG1 monoclonal antibody that specifically targets the p19 subunit of IL-23, a cytokine known to play a key role in driving inflammatory autoimmune diseases, such as IBD.
The clinical significance of these findings/modifications is unknown.
FcγRs=receptors for the Fc fragment of immunoglobulin G; IBD=inflammatory bowel disease; IgG=immunoglobulin G; IL-23=intereukin-23; IL-23p19=interleukin-23 subunit alpha; NOD2=nucleotide-binding oligomerization domain-containing protein 2; Th1=T helper 1 cell; Th17=T helper 17 cell.
Efficacy in Crohn’s Disease
Efficacy in Ulcerative Colitis
SKYRIZI Dosing
INDICATIONS AND IMPORTANT SAFETY INFORMATION FOR SKYRIZI® (risankizumab-rzaa)1 Indications Plaque Psoriasis: SKYRIZI is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. Psoriatic Arthritis: SKYRIZI is indicated for the treatment of active psoriatic arthritis in adults. Crohn's Disease: SKYRIZI is indicated for the treatment of moderately to severely active Crohn's disease in adults. Ulcerative Colitis: SKYRIZI is indicated for the treatment of moderately to severely active ulcerative colitis in adults. |
Important Safety Information
Hypersensitivity Reactions
SKYRIZI® (risankizumab-rzaa) is contraindicated in patients with a history of serious hypersensitivity reaction to risankizumab-rzaa or any of the excipients. Serious hypersensitivity reactions, including anaphylaxis, have been reported with the use of SKYRIZI. If a serious hypersensitivity reaction occurs, discontinue SKYRIZI and initiate appropriate therapy immediately.
Infection
SKYRIZI may increase the risk of infection. Do not initiate treatment with SKYRIZI in patients with a clinically important active infection until it resolves or is adequately treated.
In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing SKYRIZI. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur. If a patient develops such an infection or is not responding to standard therapy, closely monitor and discontinue SKYRIZI until the infection resolves.
Tuberculosis (TB)
Prior to initiating treatment with SKYRIZI, evaluate for TB infection and consider treatment in patients with latent or active TB for whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during and after SKYRIZI treatment. Do not administer SKYRIZI to patients with active TB.
Hepatotoxicity in Treatment of Inflammatory Bowel Disease
Drug-induced liver injury was reported in a patient with Crohn’s disease who was hospitalized for a rash during induction dosing of SKYRIZI. For the treatment of Crohn's disease and ulcerative colitis, evaluate liver enzymes and bilirubin at baseline and during induction (12 weeks); monitor thereafter according to routine patient management. Consider an alternate treatment for patients with evidence of liver cirrhosis. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct your patient to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfunction.
Administration of Vaccines
Avoid use of live vaccines in patients treated with SKYRIZI. Medications that interact with the immune system may increase the risk of infection following administration of live vaccines. Prior to initiating SKYRIZI, complete all age-appropriate vaccinations according to current immunization guidelines.
Adverse Reactions
Most common (≥1%) adverse reactions associated with SKYRIZI in plaque psoriasis and psoriatic arthritis include upper respiratory infections, headache, fatigue, injection site reactions, and tinea infections.
In psoriatic arthritis phase 3 trials, the incidence of hepatic events was higher with SKYRIZI compared to placebo.
Most common (>3%) adverse reactions associated with SKYRIZI in Crohn’s disease are upper respiratory infections, headache, and arthralgia in induction and arthralgia, abdominal pain, injection site reactions, anemia, pyrexia, back pain, arthropathy, and urinary tract infection in maintenance.
Most common (≥3%) adverse reactions associated with SKYRIZI in ulcerative colitis are arthralgia in induction, and arthralgia, pyrexia, injection site reactions, and rash in maintenance.
Lipid Elevations: Increases from baseline and increases relative to placebo were observed at Week 4 and remained stable to Week 12 in patients treated with SKYRIZI in Crohn’s disease. Lipid elevations observed in patients with ulcerative colitis were similar to those in Crohn's disease.
Dosage Forms and Strengths: SKYRIZI (risankizumab-rzaa) is available in a 150 mg/mL prefilled syringe and pen, a 600 mg/10 mL single-dose vial for intravenous infusion, and a 180 mg/1.2 mL or 360 mg/2.4 mL single-dose prefilled cartridge with on-body injector.
INDICATIONS
Plaque Psoriasis: SKYRIZI is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
Psoriatic Arthritis: SKYRIZI is indicated for the treatment of active psoriatic arthritis in adults.
Crohn's Disease: SKYRIZI is indicated for the treatment of moderately to severely active Crohn's disease in adults.
Ulcerative Colitis: SKYRIZI is indicated for the treatment of moderately to severely active ulcerative colitis in adults.
Please see Full Prescribing Information.
US-SKZG-240258
REFERENCES
1. SKYRIZI [package insert]. North Chicago, IL: AbbVie Inc.
2. Data on file, AbbVie Inc. Source: Symphony Health, an ICON plc Company, IDV®; June 2024–March 2025.
3. Data on file, AbbVie Inc. ABVRRTI81029.
4. Data on file, AbbVie Inc. ABVRRTI80749.
5. Data on file, AbbVie Inc. Source: Symphony Health, an ICON plc Company, IDV®; June 1, 2024–March 31, 2025.
6. Data on file, AbbVie Inc. #1 1L biologic badge. 2025.
7. Data on file, AbbVie Inc. Source: IQVIA (NPA, NSP) and Symphony Health data through October 2023.
8. Hawkes JE, Chan TC, Krueger JG. Psoriasis pathogenesis and the development of novel targeted immune therapies. Allergy Clin Immunol. 2017;140(3):645-653.
9. Neurath MF. IL-23: a master regulator in Crohn disease. Nat Med. 2007;13(1):26-28.
10. Zhou L, Wang Y, Wan Q, et al. A non-clinical comparative study of IL-23 antibodies in psoriasis. MAbs. 2021;13(1):1964420.