EFFICACY DATA FOR CROHN'S DISEASE
Proven to Give Patients a Chance at Endoscopic Control and Durable Remission in Crohn's1,2

||Endoscopic Remission at Week 52 was not statistically significant under the prespecified multiple testing procedure.
¶Week 152 data is based on not statistically significant, as-observed data. No conclusions can be made.
STUDY DESIGNS
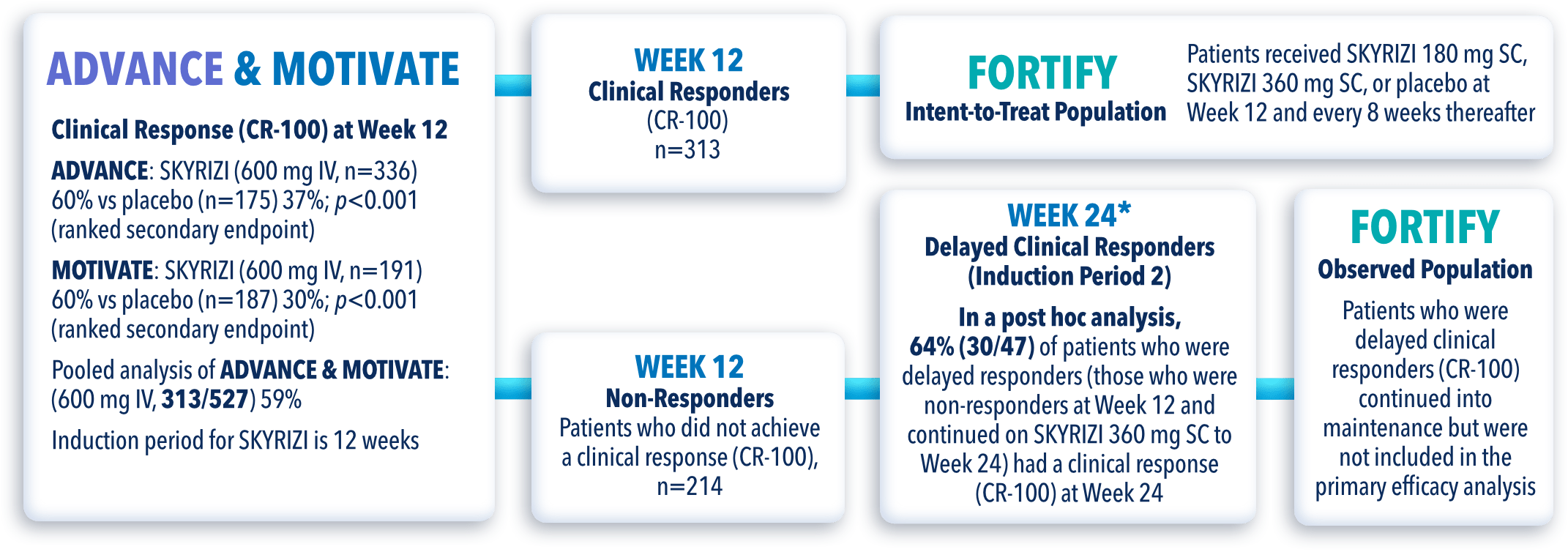
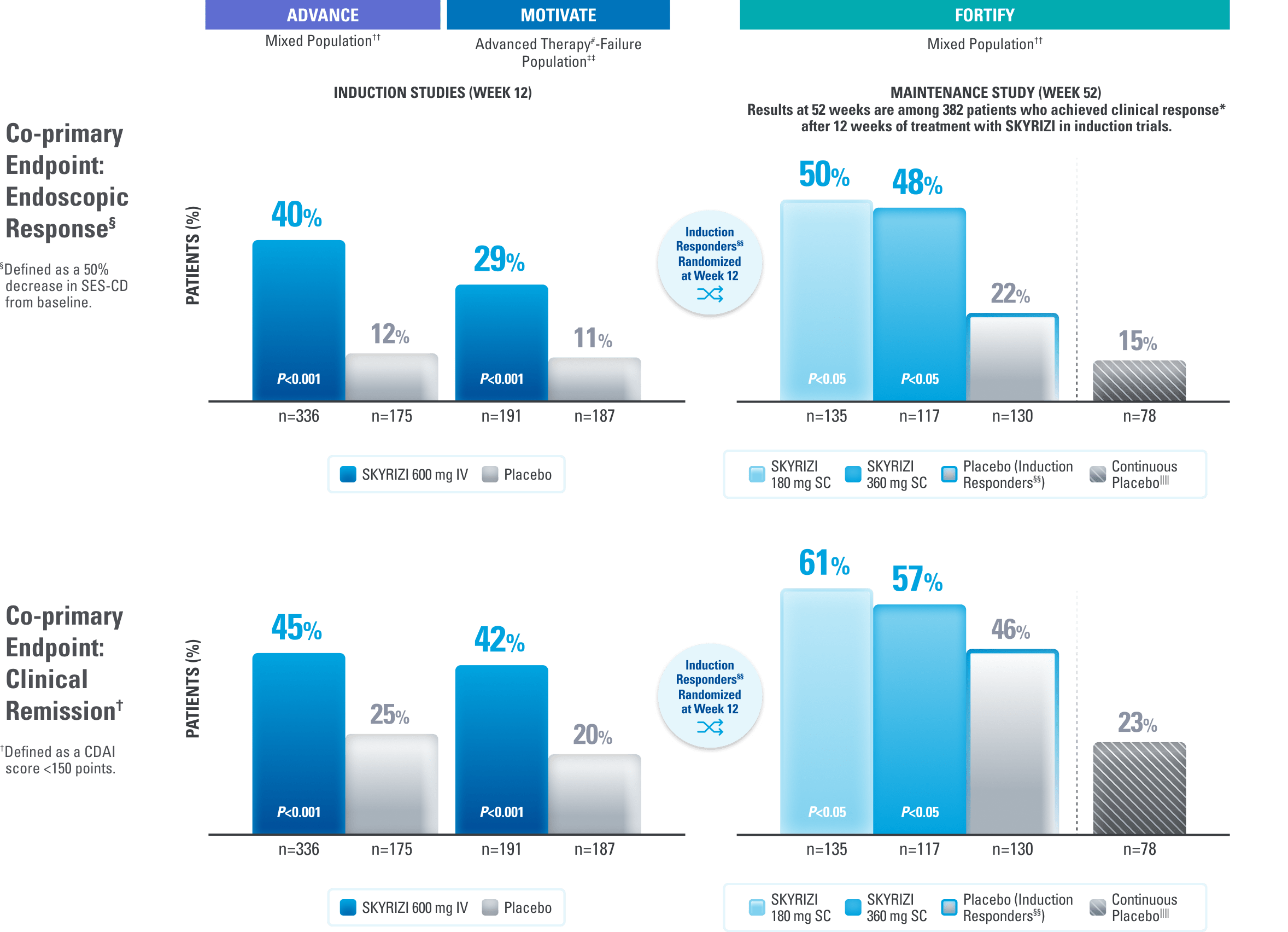
ADVANCE (N=850) and MOTIVATE (N=569) Induction studies were 12-week, randomized, double-blind, placebo-controlled studies that evaluated the efficacy and safety of SKYRIZI in patients with moderately to severely active Crohn’s disease who demonstrated prior treatment failure to conventional (AT-naïve#**) and/or biologic treatment. Patients received an IV infusion of SKYRIZI 600 mg (recommended dose), risankizumab-rzaa 1200 mg, or placebo at Weeks 0, 4, and 8.1,3
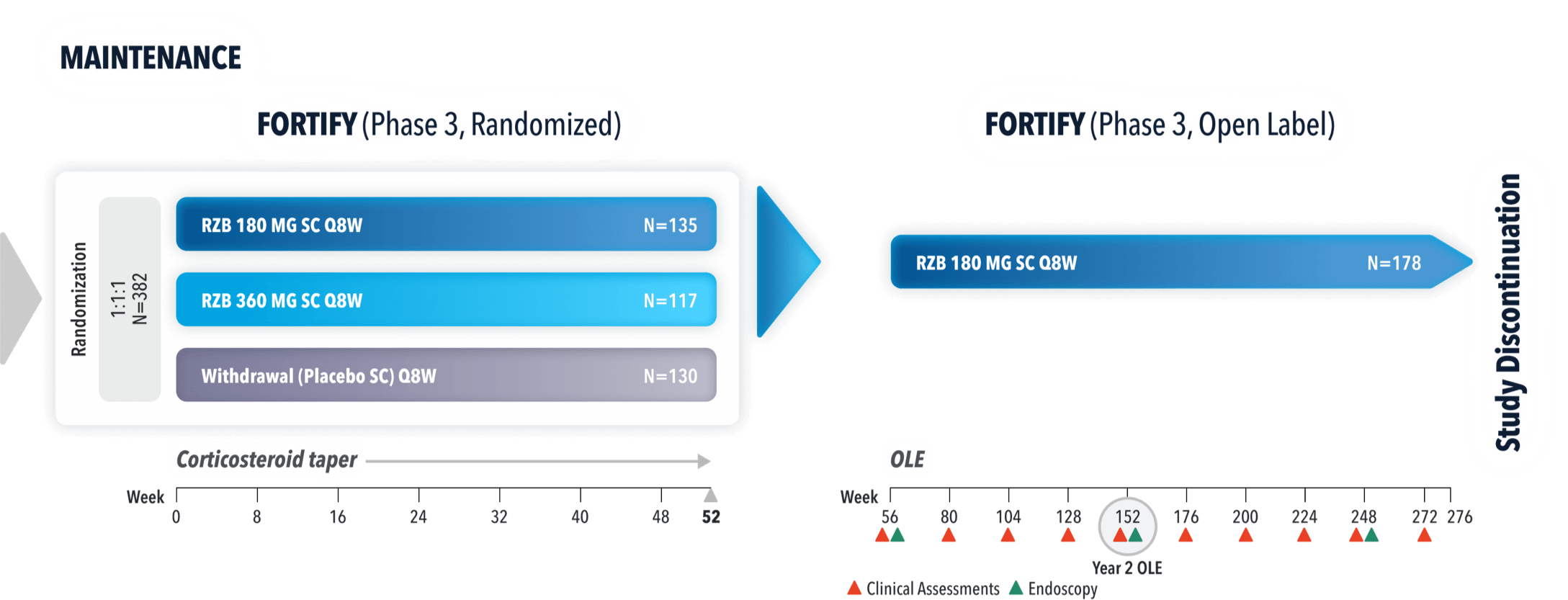
FORTIFY (N=382) Maintenance study was a 52-week study that evaluated the efficacy and safety of SKYRIZI in patients who achieved clinical response* (decrease in CDAI ≥100) from SKYRIZI induction in the ADVANCE and MOTIVATE studies. Patients were randomized to SKYRIZI 180 mg SC, SKYRIZI 360 mg SC, or placebo at Week 12 and every 8 weeks thereafter.1,4
Patients Achieved Visible Improvement of the Intestinal Lining and Clinical Remission† at Weeks 12 and 521,4
Use the lowest effective dosage to maintain therapeutic response.
All P-values are SKYRIZI treatment arms vs placebo.
IIIContinuous placebo data not intended for direct comparison.
#Advanced therapy in Crohn’s is defined as biologics.1
§§Placebo (Induction Responders): Patients who achieved CDAI clinical response (CR-100)* to SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.1
1200 mg is NOT an FDA-approved dosing regimen. The 1200 mg dosage did not demonstrate additional treatment benefit over the 600 mg dosage and is not a recommended regimen.1
*Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
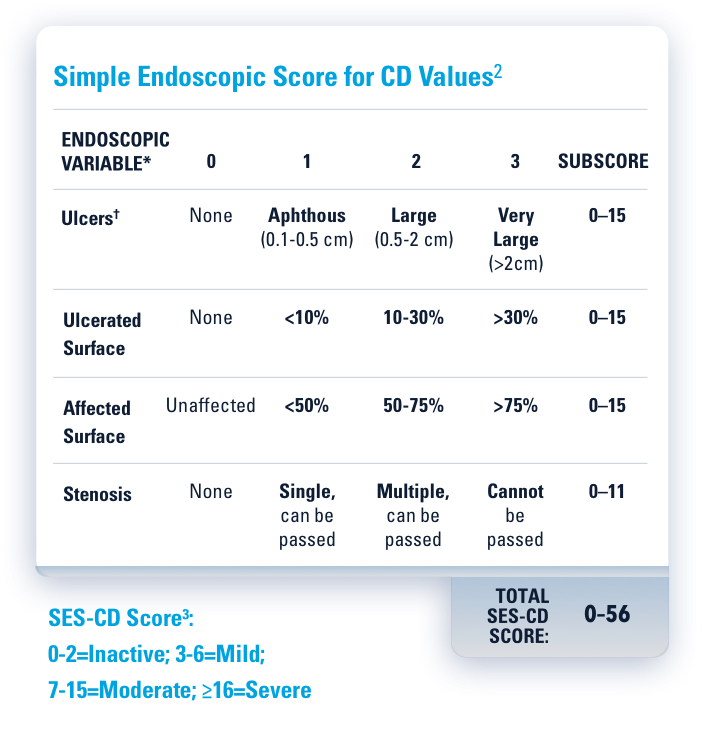
‡Endoscopic remission was defined as a SES-CD ≤4 and at least a 2-point reduction versus baseline and no subscore >1 in any individual variable, as scored by a central reviewer.1
**The advanced therapy-naïve subpopulation includes patients who were exposed to a biologic but did not have an inadequate response, loss of response, or intolerance to biologics.4
††The mixed population includes patients who had inadequate response, loss of response, or intolerance to one or more biologics (advanced therapy failure), as well as patients who were bio-exposed but did not have an inadequate response, loss of response, or intolerance to biologics (biologic naïve).1
‡‡Prior advanced therapy failure includes inadequate response, loss of response, or intolerance to one or more biologics.1
AT=advanced therapy; CDAI=Crohn's disease activity index; CR=clinical response; IV=intravenous; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease.
EARLY SYMPTOM RELIEF
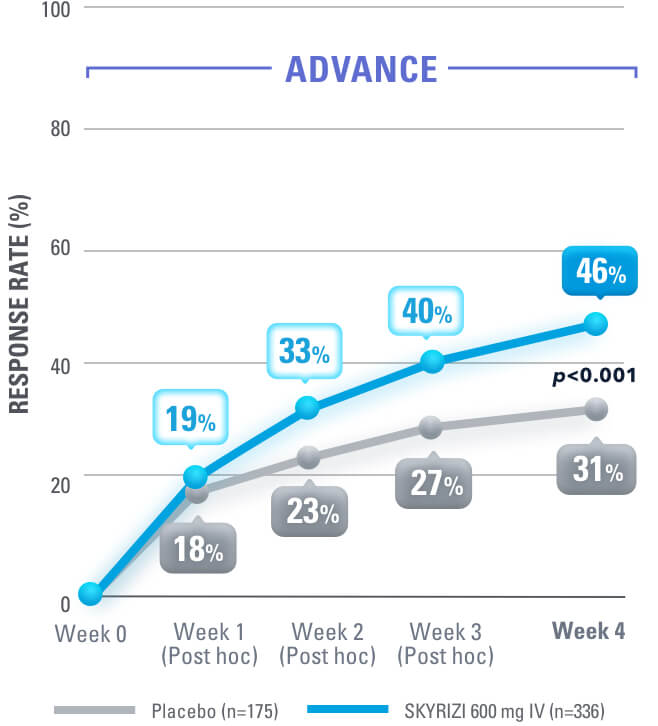
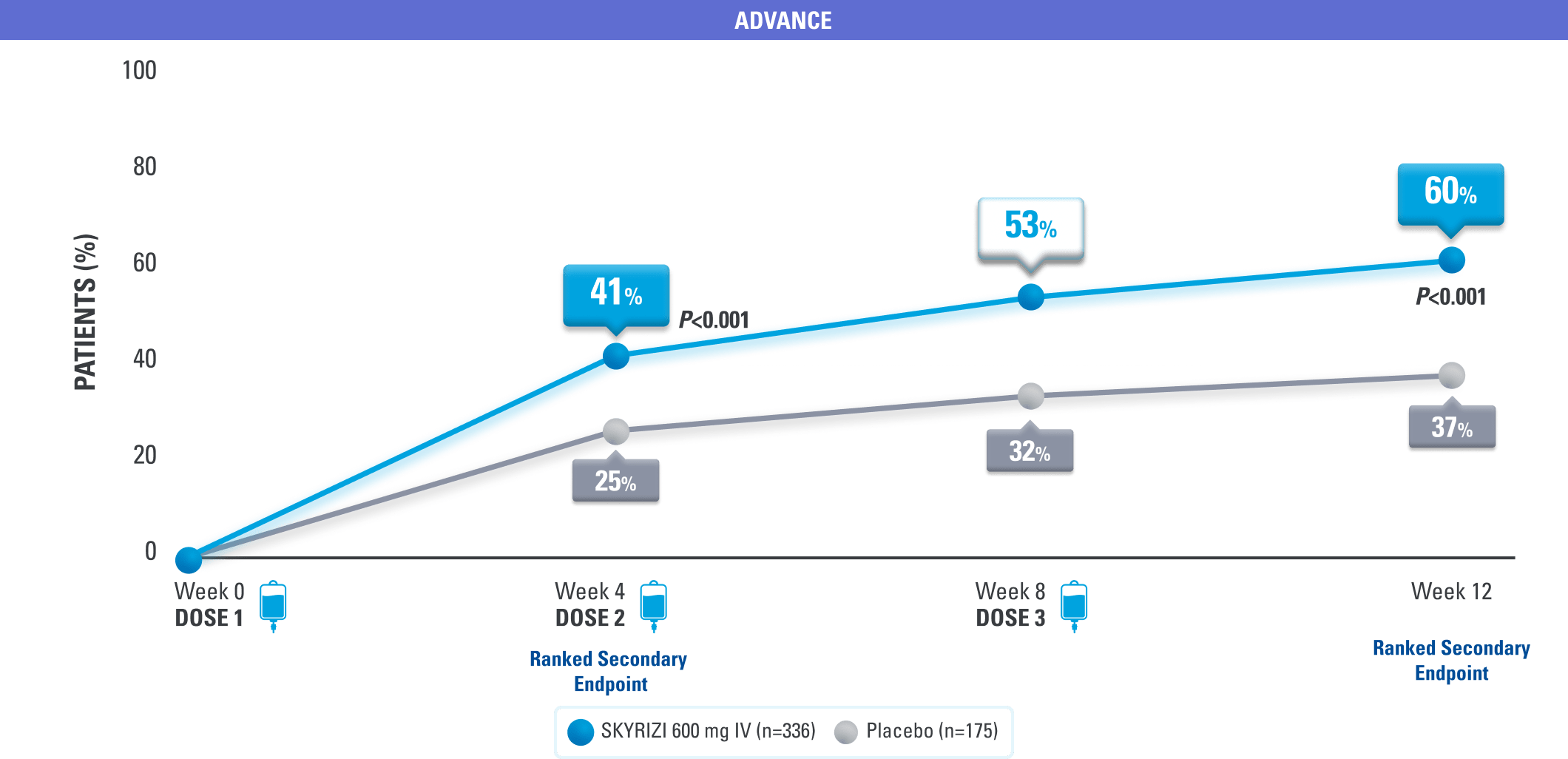
SKYRIZI Is the Only IL-23i to Achieve Symptom Relief at Week 4 in Crohn’s Pivotal Trials1,3
Clinical Response* (CDAI CR-100) at Weeks 4, 8, 121,3
DATA LIMITATIONS: Data not labeled as a ranked secondary endpoint were prespecified nonranked endpoints not controlled for multiplicity; therefore, treatment differences could represent chance findings. No conclusions regarding these comparisons can be made.
*Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
Remission as Early as Week 4
Clinical Remission at Weeks 4, 8, 125
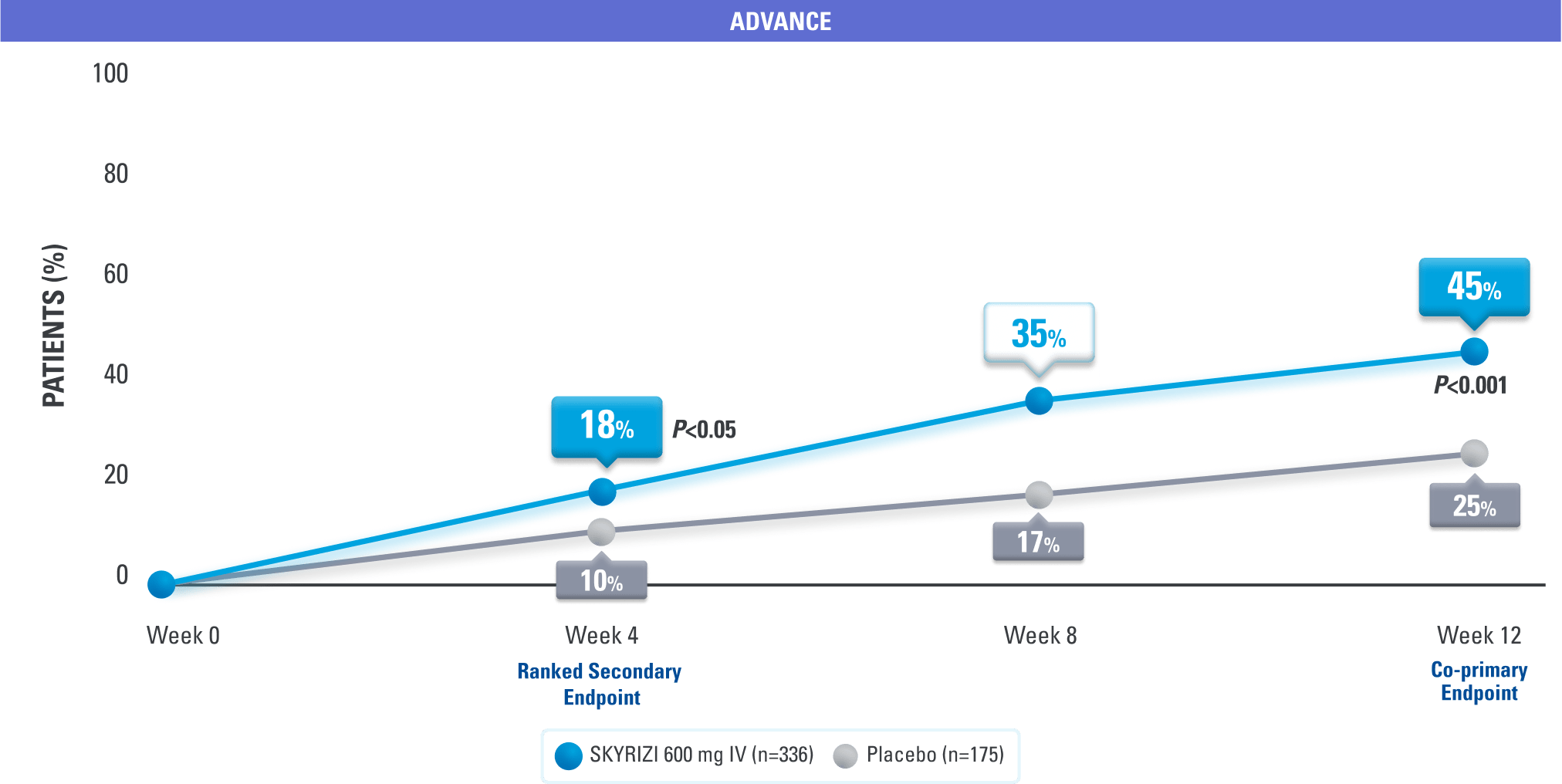

DATA LIMITATIONS: Data at Week 8 were nonranked endpoints not controlled for multiplicity; therefore, treatment differences could represent chance findings. No conclusions regarding these comparisons can be made.

SKYRIZI is the only IL-23–specific inhibitor that achieved clinical remission† at Week 4 in Crohn’s disease pivotal trials3
†Clinical remission was defined as a CDAI score <150 points.1
CDAI=Crohn's disease activity index; CR=clinical response; IV=intravenous.
OPEN-LABEL EXTENSION (OLE) STUDY DESIGN
FORTIFY-OLE is an ongoing, multicenter, open-label extension study that evaluated long-term efficacy and safety of SKYRIZI that included patients from the FORTIFY maintenance study. Patients in the FORTIFY maintenance study achieved clinical response* with SKYRIZI in the ADVANCE and MOTIVATE induction studies. This study population for FORTIFY-OLE is a sub-analysis that includes all randomized subjects who had a baseline eligibility SES-CD of ≥6 (≥4 for isolated ileal disease), received 600 mg IV SKYRIZI for 12 weeks in the induction studies, and have received at least one dose of SKYRIZI 180 mg or 360 mg SC in the Phase 3 maintenance study FORTIFY and at least one dose of SKYRIZI 180 mg SC in the FORTIFY-OLE study.1,2
ENDOSCOPIC CONTROL
Endoscopic Control: Remission† Seen at Weeks 12, 52, and 3 Years1,4,6

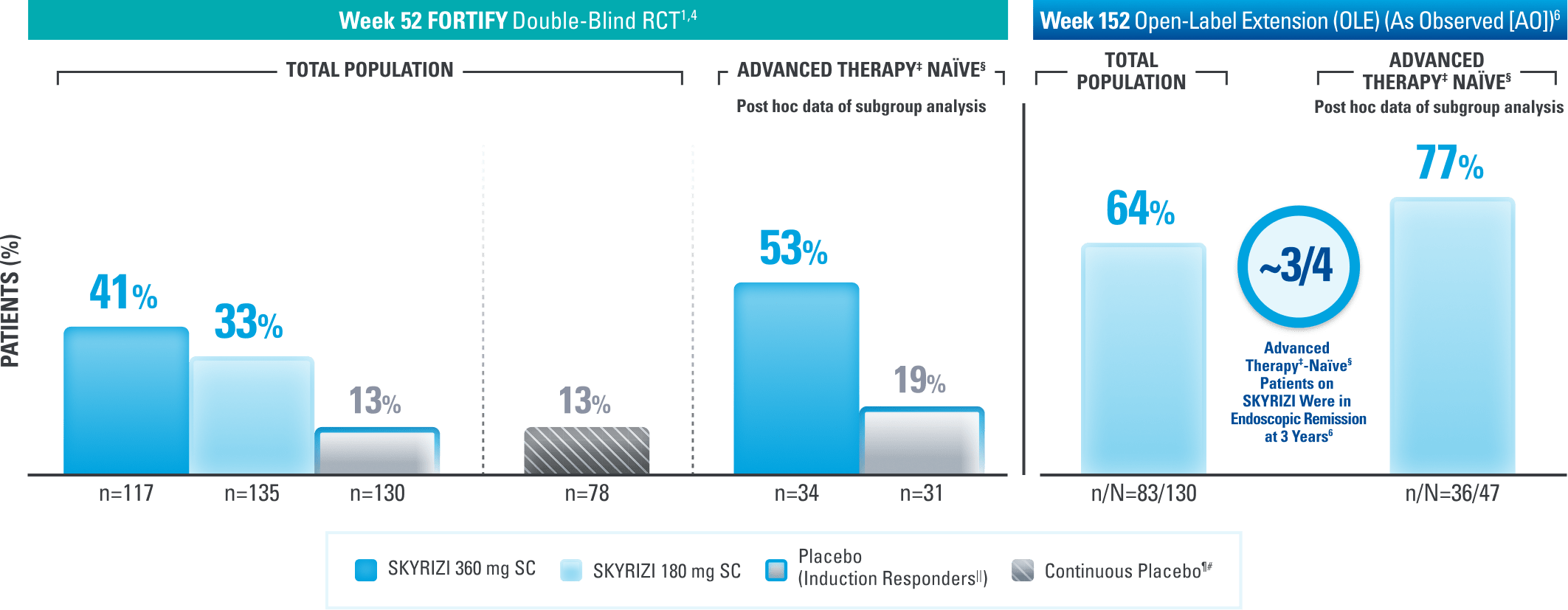
Results at 52 weeks are among 382 patients who achieved clinical response* after 12 weeks of treatment with SKYRIZI in induction trials.
Use the lowest effective dosage to maintain therapeutic response.
#Continuous placebo data not intended for direct comparison.4
DATA LIMITATIONS: Endoscopic remission at Week 52 was not statistically significant under the prespecified testing procedure. Subgroup data were not tested for multiplicity control, so no statistically significant difference in treatment effect for SKYRIZI vs placebo can be concluded.
LIMITATIONS: OLE (at Week 152): All patients completed the 52-week maintenance period and received SKYRIZI 180 mg Q8W regardless of the maintenance dose. An OLE may enrich long-term data as patients intolerant or unresponsive to the drug drop out. At Week 152, endoscopy scores were locally read. AO: A patient may not have had a response at each timepoint and any missing visit data were excluded from calculations for that visit, which may increase the percent responders. All observed data were used regardless of premature discontinuation of study drug or initiation of concomitant medication.
*Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
†Endoscopic remission was defined as SES-CD ≤4 and at least a 2-point reduction versus baseline and no subscore >1 in any individual variable, as scored by a central reviewer.1 At Week 152, endoscopy scores were locally read.
‡Advanced therapy in Crohn’s is defined as biologics.
§The advanced therapy-naïve subpopulation includes patients who were exposed to a biologic but did not have an inadequate response, loss of response, or intolerance to advanced therapies.4
||Placebo (Induction Responders): Patients who achieved CDAI clinical response (CR-100)* to SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.1
¶Patients who responded to placebo in induction (CR-100)* were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.4
AT=advanced therapy; CDAI=Crohn's disease activity index; CR=clinical response; IV=intravenous; RCT=randomized controlled trial; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease.
Endoscopic Control: Visible Improvement of the Intestinal Lining at 1 Year and Seen Up to ~3 Years1,4,6
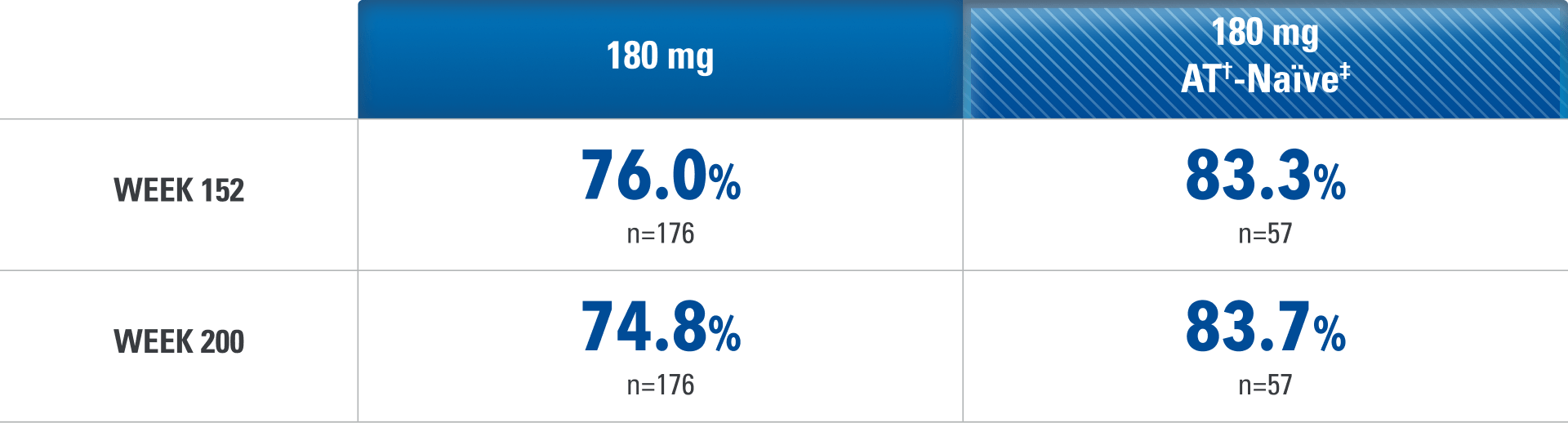
Endoscopic Response* at Weeks 52 and 1521,4,6
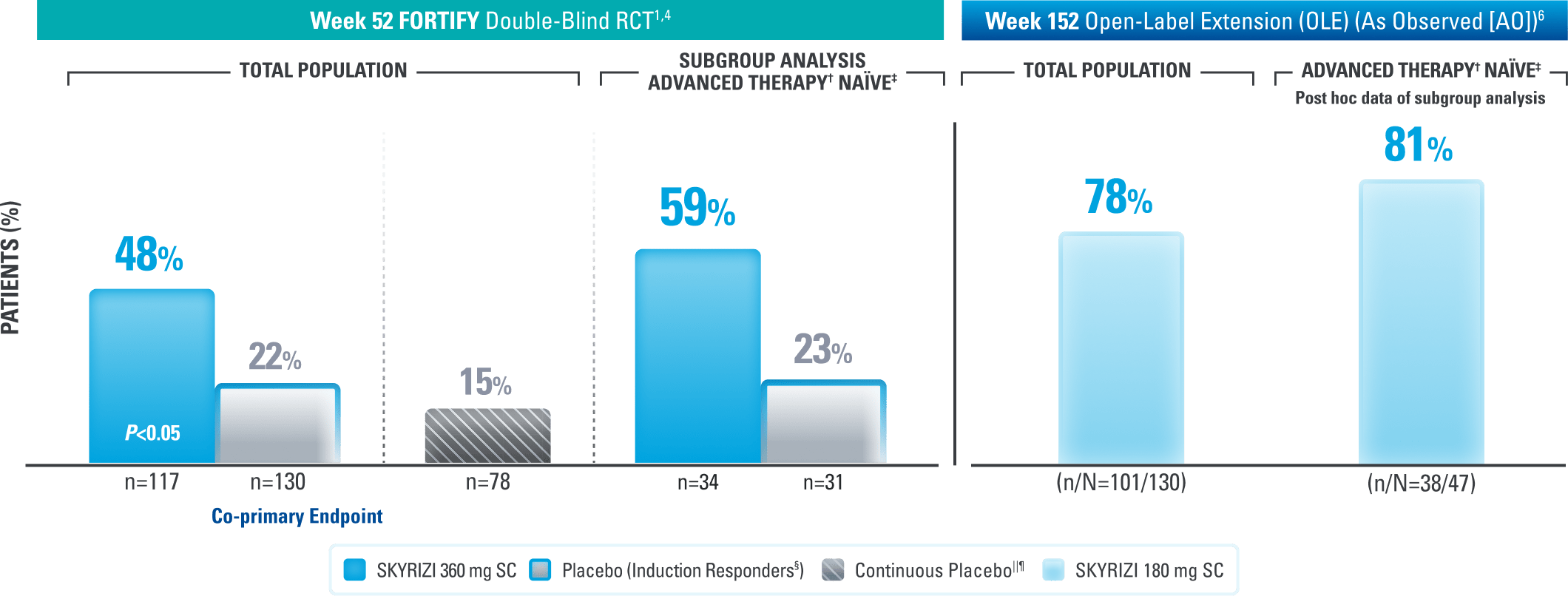
Results at 52 weeks are among 382 patients who achieved clinical response# after 12 weeks of treatment with SKYRIZI in induction trials.
Use the lowest effective dosage to maintain therapeutic response.
||Continuous placebo data not intended for direct comparison.4
LIMITATIONS: OLE: All patients completed the 52-week maintenance period and received SKYRIZI 180 mg Q8W regardless of the maintenance dose. An OLE may enrich long-term data as patients intolerant or unresponsive to the drug drop out. At Week 152, endoscopy scores were locally read. AO: A patient may not have had a response at each timepoint and any missing visit data were excluded from calculations for that visit, which may increase the percent responders. All observed data were used regardless of premature discontinuation of study drug or initiation of concomitant medication.
*Endoscopic response was defined as a decrease in SES-CD >50% from baseline, or a decrease of at least 2 points for subjects with a baseline score of 4 and isolated ileal disease, based on central reading. The sections evaluated on endoscopy are the rectum, sigmoid and left colon, transverse colon, right colon, and ileum (per SES-CD assessment).1
†Advanced therapy in Crohn’s is defined as biologics.
‡The advanced therapy-naïve subpopulation includes patients who were exposed to a biologic but did not have an inadequate response, loss of response, or intolerance to advanced therapies.4
§Placebo (Induction Responders): Patients who achieved CDAI clinical response (CR-100)# to SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.1
¶Patients who responded to placebo in induction (CR-100)# were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis. 4
#Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
CDAI=Crohn's disease activity index; CR=clinical response; IV=intravenous; RCT=randomized controlled trial; SC=subcutaneous; SES-CD=simple endoscopic score for Crohn's disease.
DURABLE REMISSION
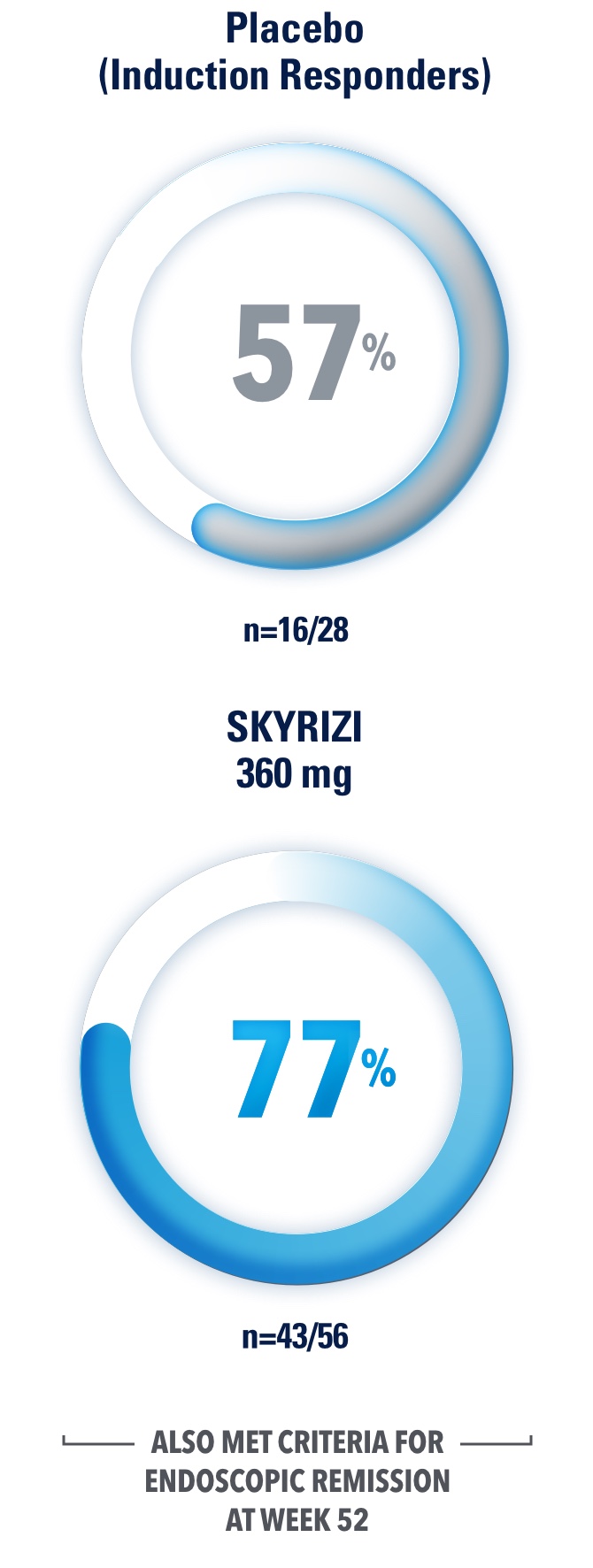
Durable Remission at 1 Year1,3,4
Clinical Remission* at Week 521,3,4
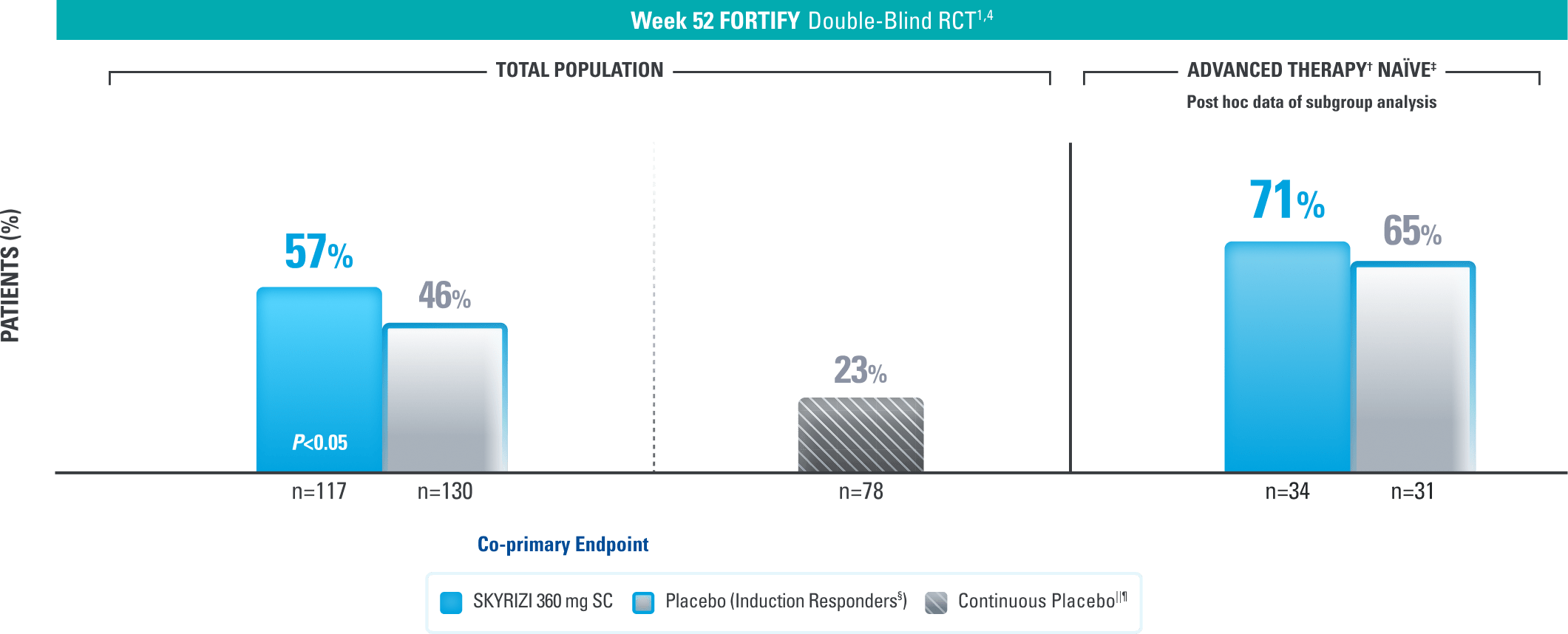
Results at 52 weeks are among 382 patients who achieved clinical response# after 12 weeks of treatment with SKYRIZI in induction trials.
||Continuous placebo data not intended for direct comparison.4
*Clinical remission is defined as a CDAI score <150 points.1
†Advanced therapy in Crohn’s is defined as biologics.
‡The advanced therapy-naïve subpopulation includes patients who were exposed to a biologic but did not have an inadequate response, loss of response, or intolerance to advanced therapies.4
§Placebo (Induction Responders): Patients who achieved CDAI clinical response (CR-100)# to SKYRIZI induction therapy and were randomized to receive placebo in the maintenance study.1
¶Patients who responded to placebo in induction (CR-100)# were not randomized and continued on placebo in maintenance. These patients were not included in the primary efficacy analysis.4
#Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
CDAI=Crohn's disease activity index; CR=clinical response; Q8W=every 8 weeks; RCT=randomized controlled trial; SC=subcutaneous.
Consistent Rates of Clinical Remission* Up to ~4 Years1,2,6
Data Up to ~4 Years Based on a FORTIFY Post Hoc Analysis (As Observed [AO])2,6
Clinical response† to SKYRIZI required after 12 weeks of induction to enter for FORTIFY1
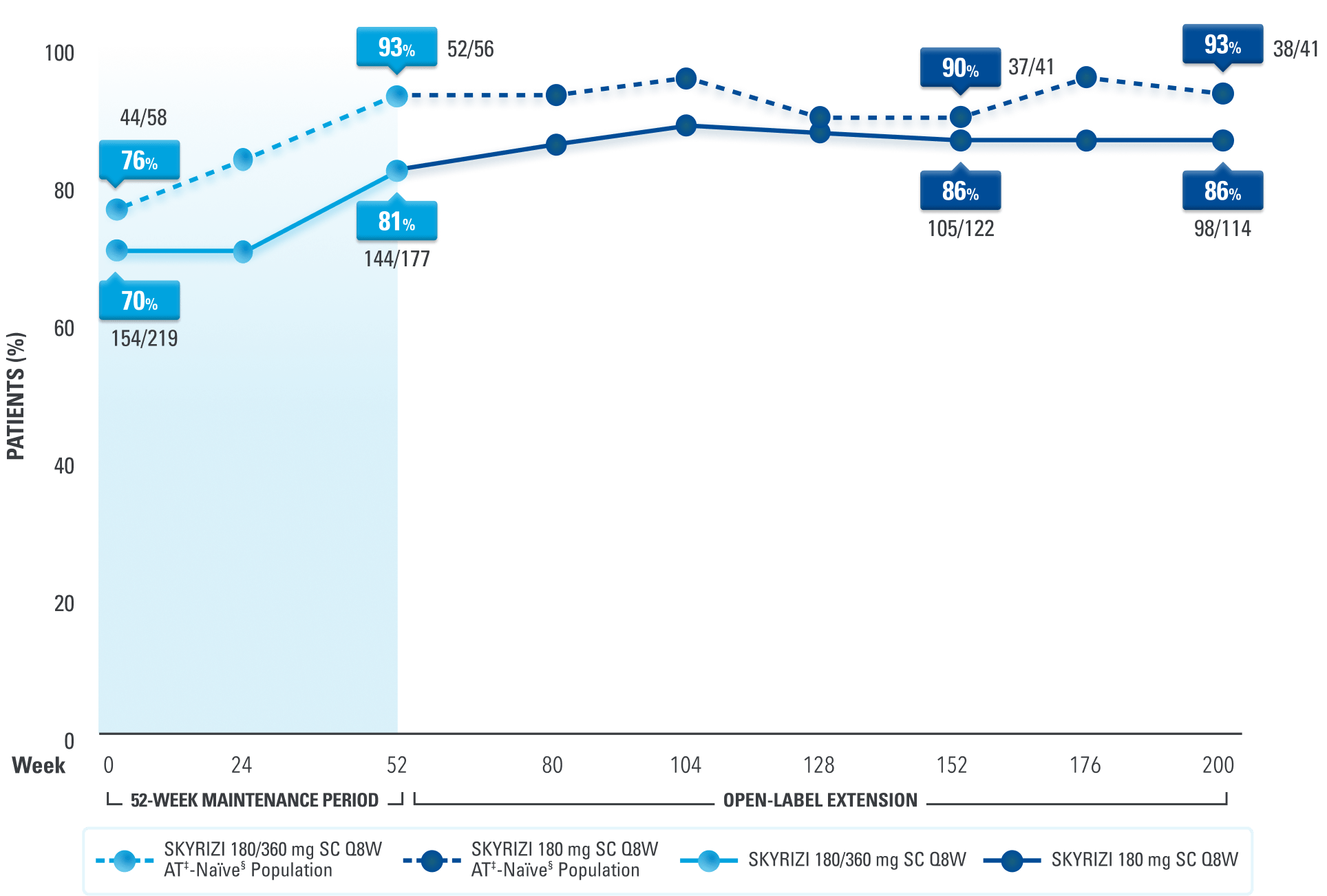
of AT‡-naïve§ patients who entered the OLE experienced clinical remission* at ~4 years6
LIMITATIONS: OLE: All patients completed the 52-week maintenance period and received SKYRIZI 180 mg Q8W regardless of the maintenance dose. An OLE may enrich long-term data as patients intolerant or unresponsive to the drug drop out. AO: A patient may not have had a response at each timepoint and any missing visit data were excluded from calculations for that visit, which may increase the percent responders. All observed data were used regardless of premature discontinuation of study drug or initiation of concomitant medication.
DATA LIMITATIONS for Post Hoc Analysis: A post hoc analysis is not adjusted for multiplicity; thus, no statistical inferences can be made due to the exploratory nature of the analysis.
§The advanced therapy-naïve subpopulation includes patients who were exposed to a biologic but did not have an inadequate response, loss of response, or intolerance to advanced therapies.4
*Clinical remission is defined as a CDAI score <150 points.1
†Clinical response was defined as a reduction of CDAI score ≥100 points from baseline.1
‡Advanced therapy in Crohn’s is defined as biologics.
CDAI=Crohn's disease activity index; CR=clinical response; OLE=open-label extension; Q8W=every 8 weeks; RCT=randomized controlled trial; SC=subcutaneous.