Over a 3-year period, SKYRIZI Q8W maintenance dosing with the OBI could result in ~20 to 60 fewer injections compared to IL-23i Q4W maintenance dosing treatment options.1-3
DOSING
FOR SKYRIZI
Consistent Dosing Your Patients Can Count On
DOSING SCHEDULE
Reliable 2-Month Maintenance Dosing After 3 IV Infusions for Crohn’s Disease and Ulcerative Colitis1
Maintenance doses administered in the office or at home1
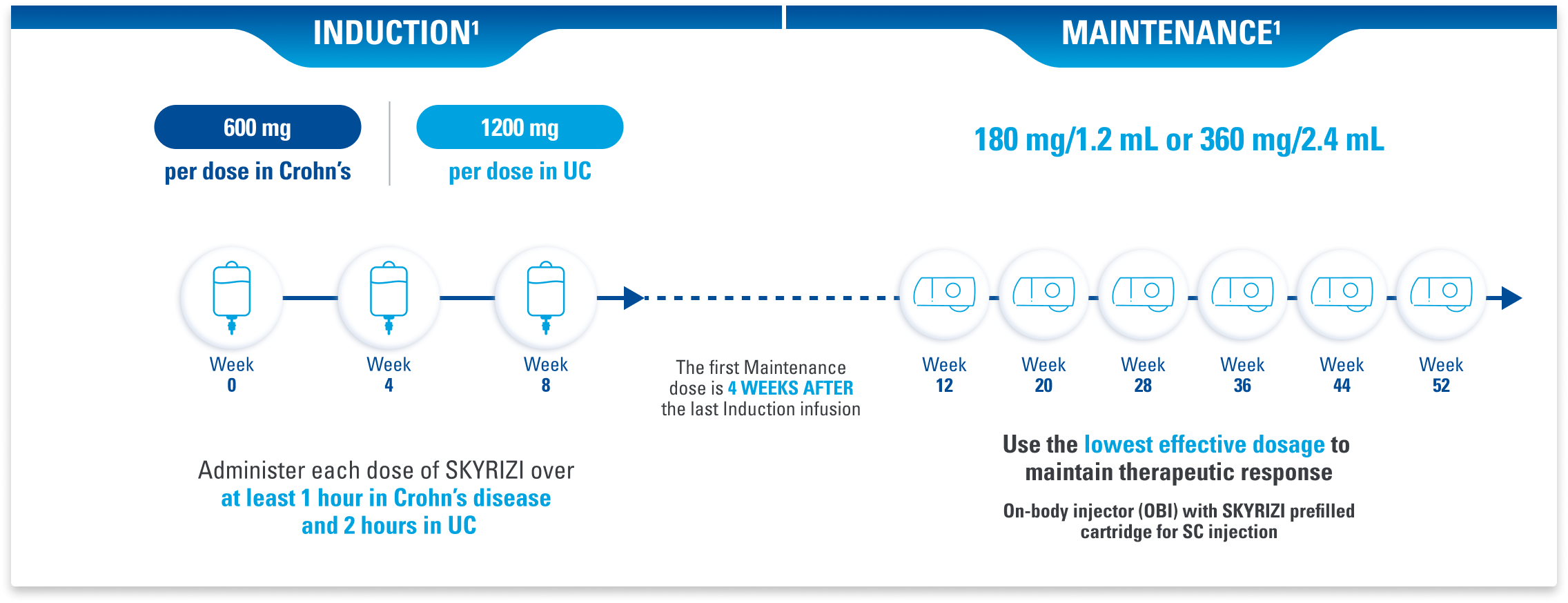
ADMINISTRATION CONSIDERATIONS: SKYRIZI is intended for use under the guidance and supervision of a healthcare professional (HCP). SKYRIZI vial for intravenous administration is intended for administration by an HCP. Prior to starting therapy, please refer to the Dosage and Administration section of the Prescribing Information for complete information on how to initiate, prepare, and administer SKYRIZI. Patients may self-inject SKYRIZI using the on-body injector with prefilled cartridge after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI according to the Instructions for Use.1
Swipe to scroll
| LAB MONITORING Treatment Considerations in Crohn's Disease and UC1 | ||
|---|---|---|
| Perform lab tests for1: Liver enzymes,* bilirubin | ||
| Check lab values | Treatment should not be initiated or continued if: | Additional considerations |
| Evaluate at baseline and during induction (for at least 12 weeks). Monitor thereafter according to routine patient management. | Drug-induced liver injury is suspected, until this diagnosis is excluded. |
Consider other treatment options in patients with evidence of liver cirrhosis. Prompt investigation of the cause of liver enzyme elevation is recommended to identify potential cases of drug-induced liver injury. |
Infections: SKYRIZI may increase the risk of infections. Treatment with SKYRIZI should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur. If such an infection develops, monitor the patient closely and do not administer SKYRIZI until the infection resolves.
Tuberculosis (TB): Evaluate for TB infection prior to initiating treatment with SKYRIZI.
Vaccinations: Complete all age-appropriate vaccinations according to current immunization guidelines prior to initiating treatment with SKYRIZI.
*Liver enzymes include ALT and AST.
ALT=alanine aminotransferase; AST=aspartate aminotransferase; IV=intravenous; SC=subcutaneous.

Dosing and Monitoring Guide
Quick access to dosing- and monitoring-related information as you get your patients started on SKYRIZI.
IL-23i DOSING
SKYRIZI Offers the Same Maintenance Schedule and OBI Device, Regardless of Indication or Dosage Strength1-3
Dosing Schedules for IL-23 Inhibitors in Adults With Moderate to Severe Crohn’s Disease1-3

*Inject 300 mg subcutaneously (given as 2 consecutive injections of 100 mg and 200 mg in any order).
IL-23=interleukin-23; IL-23i=interleukin-23 inhibitor; OBI=on-body injector; Q4W=every 4 weeks; Q8W=every 8 weeks.
Dosing Schedules for IL-23 Inhibitors in Adults With Moderate to Severe UC1-3
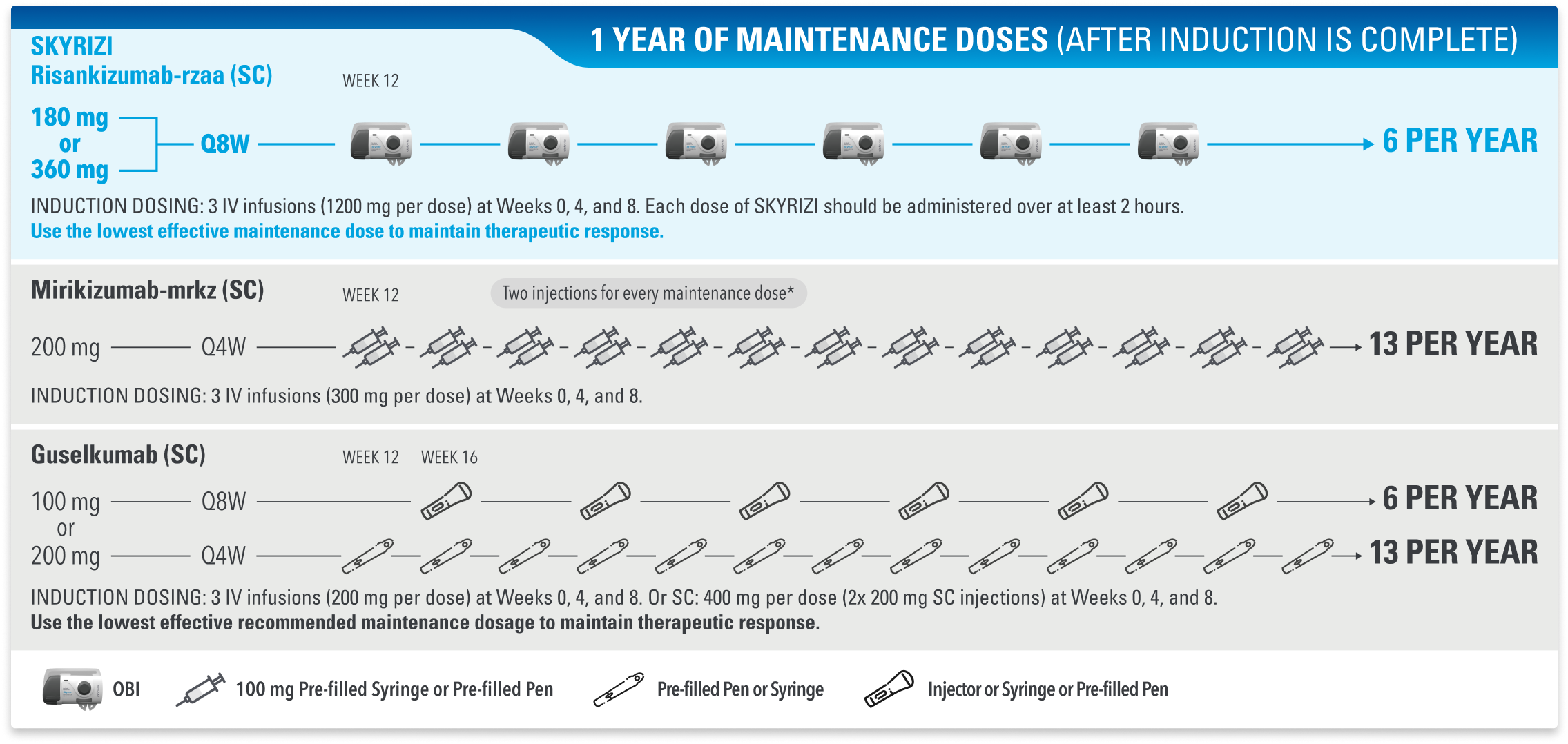
*Inject 200 mg subcutaneously (given as 2 consecutive injections of 100 mg each).
IL-23=interleukin-23; IL-23i=interleukin-23 inhibitor; OBI=on-body injector; Q4W=every 4 weeks; Q8W=every 8 weeks.

This presentation is not intended to compare the efficacy or safety of the treatments shown. While these factors are important, there are additional considerations for selecting a treatment. Please refer to each product’s Prescribing Information for additional information.
Injection site reaction was the most common adverse reaction reported during SKYRIZI Crohn's and UC maintenance trials.
LAB MONITORING: Evaluate liver enzymes and billirubin at baseline and during induction (12 weeks); monitor thereafter according to routine patient management. Consider an alternate treatment for patients with evidence of liver cirrhosis. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded.
ADMINISTRATION CONSIDERATIONS: SKYRIZI is intended for use under the guidance and supervision of a healthcare professional (HCP). SKYRIZI vial for intravenous administration is intended for administration by an HCP. Prior to starting therapy, please refer to the Dosage and Administration section of the Prescribing Information for complete information on how to initiate, prepare, and administer SKYRIZI. Patients may self-inject SKYRIZI using the on-body injector with prefilled cartridge after training in subcutaneous injection technique. Provide proper training on all the required steps to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI according to the Instructions for Use.1
Recommended for You
On-Body Injector
SKYRIZI Access
Support for Your Office and Patients



