EARLY AND DURABLE SYMPTOM CONTROL
SKYRIZI ACHIEVED ITS CO-PRIMARY ENDPOINT OF CLINICAL REMISSION AT WEEK 12 AND WEEK 521-3
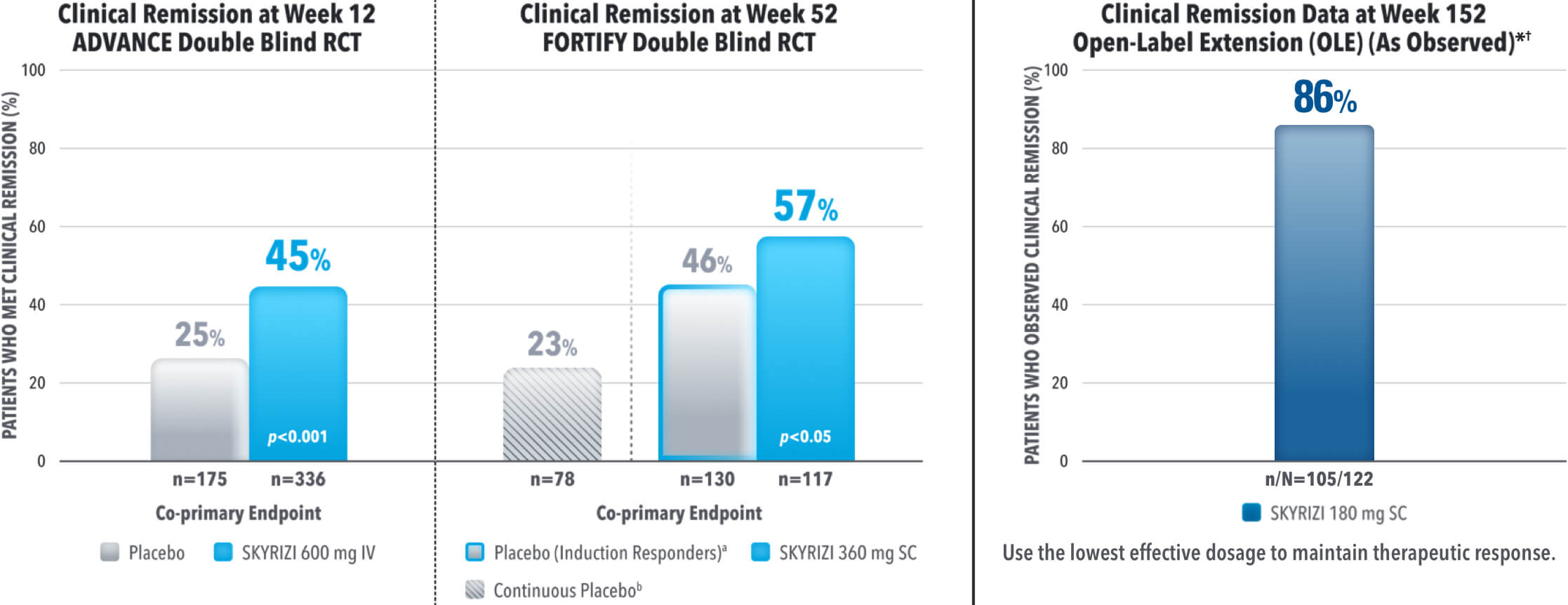
aThe induction-only group consisted of subjects who achieved clinical response (CR-100) to SKYRIZI induction therapy and were randomized to receive placebo in FORTIFY. The mean half-life of SKYRIZI is approximately 21 days for patients with Crohn's which may have contributed to these rates.
Results at 52 weeks are among 382 patients who achieved clinical response‡ after 12 weeks of treatment with SKYRIZI in induction trials.
bContinuous placebo data not intended for direct comparison.
*OLE Limitations: In an OLE, there is a potential for enrichment of the long-term data in the remaining patient populations since patients who are unable to tolerate or do not respond to the drug often drop out.
†AO Disclosure: In an as observed (AO) analysis, missing visit data was excluded from calculations for that visit, which may increase the percent of responders. All observed data was used regardless of premature discontinuation of study drug, or initiation of concomitant medication. The same patient may not have a response at each timepoint.